Shield Therapeutics PLC Notice of Results (2111I)
11 Agosto 2021 - 1:00AM
UK Regulatory
TIDMSTX
RNS Number : 2111I
Shield Therapeutics PLC
11 August 2021
Shield Therapeutics plc
("Shield or the "Company" or the "Group")
Notice of Results
London, UK, 11 August 2021: Shield Therapeutics plc (LSE: STX),
a commercial stage pharmaceutical company with a focus on
addressing iron deficiency with its lead product
Feraccru(R)/Accrufer(R) (ferric maltol), will announce its interim
results for the six months ended 30 June 2021 on Tuesday 17 August
2021.
Analyst briefing
A briefing open to analysts will take place remotely via video
conference call on Tuesday 17 August 2021 at 14.00 (BST). If you
would like the details of this call please contact Walbrook PR on
shield@walbrookpr.com .
For further information, please contact:
Shield Therapeutics plc +44 (0) 191 511 8500
Greg Madison (CEO)
Hans-Peter Rudolf (CFO)
Peel Hunt LLP - Nominated Adviser & Joint
Broker +44 (0) 20 7148 8900
James Steel / Christopher Golden
finnCap Ltd - Joint Broker +44 (0) 20 7220 0500
Geoff Nash / Alice Lane/ George Dollemore
Walbrook PR - Financial PR & IR Adviser +44 (0) 20 7933 8780
Paul McManus / Lianne Cawthorne/ Alice or shield@walbrookpr.com
Woodings
About Shield
Shield is a commercial stage, pharmaceutical company with a
focus on addressing iron deficiency with its lead product Feraccru
(R) /Accrufer (R) (ferric maltol), a novel, stable, non-salt based
oral therapy for adults with iron deficiency with or without
anaemia.
Shield's lead product, Feraccru(R)/Accrufer(R), has been
approved for use in the United States, European Union, UK,
Switzerland and Australia and has exclusive IP rights until the
mid-2030s. The Group is currently launching Accrufer(R) in the US.
Feraccru(R) is already being commercialised in the UK and European
Union by Norgine B.V., who also have the marketing rights in
Australia and New Zealand. Shield also has an exclusive licence
agreement with Beijing Aosaikang Pharmaceutical Co., Ltd., for the
development and commercialisation of Feraccru(R)/Accrufer(R) in
China, Hong Kong, Macau and Taiwan.
For more information, please visit www.shieldtherapeutics.com .
Follow Shield on Twitter @ShieldTx
This information is provided by RNS, the news service of the
London Stock Exchange. RNS is approved by the Financial Conduct
Authority to act as a Primary Information Provider in the United
Kingdom. Terms and conditions relating to the use and distribution
of this information may apply. For further information, please
contact rns@lseg.com or visit www.rns.com.
RNS may use your IP address to confirm compliance with the terms
and conditions, to analyse how you engage with the information
contained in this communication, and to share such analysis on an
anonymised basis with others as part of our commercial services.
For further information about how RNS and the London Stock Exchange
use the personal data you provide us, please see our Privacy
Policy.
END
NORBLGDIDGBDGBG
(END) Dow Jones Newswires
August 11, 2021 02:00 ET (06:00 GMT)
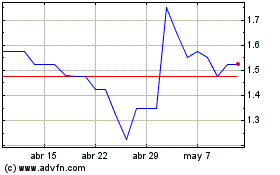
Shield Therapeutics (LSE:STX)
Gráfica de Acción Histórica
De Mar 2024 a Abr 2024
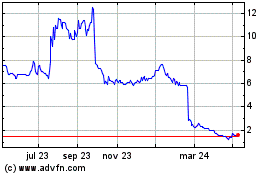
Shield Therapeutics (LSE:STX)
Gráfica de Acción Histórica
De Abr 2023 a Abr 2024