Apellis Appoints Leading Expert Dr. Philip Ferrone as Chief Medical Retina Advisor
11 Marzo 2024 - 5:00AM

Apellis Pharmaceuticals, Inc. (Nasdaq: APLS) today announced that
leading retina specialist Philip Ferrone, M.D., will join the
company as Chief Medical Retina Advisor, effective March 18.
“Phil is globally renowned for his leadership in retina,
including as a past president of the American Society of Retina
Specialists (ASRS), so we are delighted to welcome him to the
Apellis team,” said Caroline Baumal, M.D., Chief Medical Officer,
Apellis. “Phil has extensive expertise in retina research and
patient care, which will be invaluable as we continue to bring
SYFOVRE to patients with GA and advance our retina pipeline.”
“It is an honor to join Apellis in its mission to deliver
life-changing treatments for patients living with serious
diseases,” said Dr. Ferrone. “I was an early adopter of SYFOVRE and
have realized first-hand how impactful this treatment is for GA
patients. I look forward to working closely with Caroline and this
tremendously talented team to advance care for patients with this
devastating disease.”
Dr. Ferrone will continue to serve as the director of clinical
research at Vitreoretinal Consultants in New York. He is a past
president of the ASRS and has been on its board of directors for 18
years. He is also a past president of The Foundation of the ASRS,
which aims to raise patient awareness and education about retina
health.
Dr. Ferrone has served as principal investigator in over 60
clinical trials investigating new treatments for retinal diseases,
authored over 75 peer-reviewed medical manuscripts, six book
chapters, and has given over 160 scientific presentations at
national and international ophthalmology medical meetings. Dr.
Ferrone earned his undergraduate degree from Union College and his
medical degree from Harvard Medical School. He completed a
fellowship in vitreoretinal disease and surgery at Associated
Retinal Consultants in Royal Oak, MI and was Chief Resident and on
faculty at the Duke University Eye Center.
About
SYFOVRE® (pegcetacoplan
injection)SYFOVRE® (pegcetacoplan injection) is the
first-ever approved therapy for geographic atrophy (GA). By
targeting C3, SYFOVRE is designed to provide comprehensive control
of the complement cascade, part of the body’s immune system.
SYFOVRE is approved in the United States for the treatment of GA
secondary to age-related macular degeneration.
U.S. Important Safety Information for
SYFOVRE® (pegcetacoplan
injection) CONTRAINDICATIONS
- SYFOVRE is contraindicated in patients with ocular or
periocular infections, and in patients with active intraocular
inflammation
WARNINGS AND PRECAUTIONS
- Endophthalmitis and Retinal Detachments
- Intravitreal injections, including those with SYFOVRE, may be
associated with endophthalmitis and retinal detachments. Proper
aseptic injection technique must always be used when administering
SYFOVRE to minimize the risk of endophthalmitis. Patients should be
instructed to report any symptoms suggestive of endophthalmitis or
retinal detachment without delay and should be managed
appropriately.
- Retinal Vasculitis and/or Retinal Vascular Occlusion
- Retinal vasculitis and/or retinal vascular occlusion, typically
in the presence of intraocular inflammation, have been reported
with the use of SYFOVRE. Cases may occur with the first dose of
SYFOVRE and may result in severe vision loss. Discontinue treatment
with SYFOVRE in patients who develop these events. Patients should
be instructed to report any change in vision without delay.
- Neovascular AMD
- In clinical trials, use of SYFOVRE was associated with
increased rates of neovascular (wet) AMD or choroidal
neovascularization (12% when administered monthly, 7% when
administered every other month and 3% in the control group) by
Month 24. Patients receiving SYFOVRE should be monitored for signs
of neovascular AMD. In case anti-Vascular Endothelial Growth Factor
(anti-VEGF) is required, it should be given separately from SYFOVRE
administration.
- Intraocular Inflammation
- In clinical trials, use of SYFOVRE was associated with episodes
of intraocular inflammation including: vitritis, vitreal cells,
iridocyclitis, uveitis, anterior chamber cells, iritis, and
anterior chamber flare. After inflammation resolves, patients may
resume treatment with SYFOVRE.
- Increased Intraocular Pressure
- Acute increase in IOP may occur within minutes of any
intravitreal injection, including with SYFOVRE. Perfusion of the
optic nerve head should be monitored following the injection and
managed as needed.
ADVERSE REACTIONS
- Most common adverse reactions (incidence ≥5%) are ocular
discomfort, neovascular age-related macular degeneration, vitreous
floaters, conjunctival hemorrhage.
Please see accompanying full Prescribing Information for more
information.
About Apellis Apellis
Pharmaceuticals, Inc. is a global biopharmaceutical company that
combines courageous science and compassion to develop life-changing
therapies for some of the most challenging diseases patients face.
We ushered in the first new class of complement medicine in 15
years and now have two approved medicines targeting C3. These
include the first-ever therapy for geographic atrophy, a leading
cause of blindness around the world. We believe we have only begun
to unlock the potential of targeting C3 across serious retinal,
rare, and neurological diseases. For more information, please visit
http://apellis.com or follow us
on Twitter and LinkedIn.
Apellis Forward-Looking StatementStatements in
this press release about future expectations, plans and prospects,
as well as any other statements regarding matters that are not
historical facts, may constitute “forward-looking statements”
within the meaning of The Private Securities Litigation Reform Act
of 1995. The words “anticipate,” “believe,” “continue,” “could,”
“estimate,” “expect,” “intend,” “may,” “plan,” “potential,”
“predict,” “project,” “should,” “target,” “will,” “would” and
similar expressions are intended to identify forward-looking
statements, although not all forward-looking statements contain
these identifying words. Actual results may differ materially from
those indicated by such forward-looking statements as a result of
various important factors and other factors discussed in the “Risk
Factors” section of Apellis’ Annual Report on Form 10-K with the
Securities and Exchange Commission on February 27, 2024 and the
risks described in other filings that Apellis may make with the
Securities and Exchange Commission. Any forward-looking statements
contained in this press release speak only as of the date hereof,
and Apellis specifically disclaims any obligation to update any
forward-looking statement, whether as a result of new information,
future events or otherwise.Media Contact: Lissa
Pavluk media@apellis.com 617.977.6764
Investor Contact: Meredith
Kaya meredith.kaya@apellis.com617.599.8178
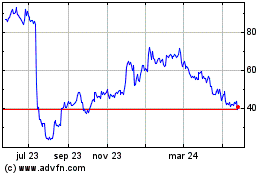
Apellis Pharmaceuticals (NASDAQ:APLS)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
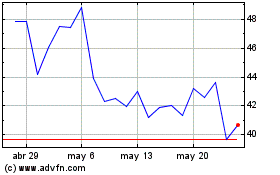
Apellis Pharmaceuticals (NASDAQ:APLS)
Gráfica de Acción Histórica
De May 2023 a May 2024