New Phase 3 data with aprocitentan to be presented at the European Society of Hypertension Annual Meeting 202
23 Junio 2023 - 10:45AM

New Phase 3 data with aprocitentan to be presented at the European
Society of Hypertension Annual Meeting 202
Allschwil, Switzerland – June
23, 2023
Idorsia Ltd (SIX: IDIA) today announced that further data for
aprocitentan, Idorsia’s investigational dual endothelin receptor
antagonist evaluating the treatment of patients with resistant
hypertension, will be presented as an oral presentation by Prof.
Markus Schlaich, MD, at the European Society of Hypertension’s 32nd
European Meeting of Hypertension and Cardiovascular Protection,
taking place in Milan, Italy, 23-26, 2023.
The presentation is scheduled for Monday, June 26 (14:05 – 14:15
CEST) in Yellow Hall 1, as part of the “Resistant and Uncontrolled
Hypertension 2” session, entitled “Effects of the dual endothelin
antagonist aprocitentan on ambulatory blood pressure indices in
patients with resistant hypertension – results from the PRECISION
study”. The presentation focuses on the effects on relevant indices
of ambulatory BP measurements (ABPM), including post-hoc analysis
of patients at high risk of cardiovascular events based on
night-time BP values. The abstract can be found in the meeting
program.
In May 2022, Idorsia announced positive top-line results of the
Phase 3 PRECISION study with aprocitentan for the
treatment of patients with resistant hypertension. Detailed results
were published in The Lancet and presented as a
Late-Breaking Science presentation during the American Heart
Association (AHA) Scientific Sessions in November 2022. More
details and commentary can be found in the dedicated press
release and an investor webcast featuring Prof.
Markus Schlaich, an investigator in PRECISION. A new drug
application (NDA) for aprocitentan was filed with the US FDA in
December 2022, and the market authorisation application (MAA) was
submitted to the EMA at the end January 2023.
Notes to the editor
About Prof. Markus Schlaich, MD
Markus Schlaich is a nephrologist and a European
Society of Hypertension (ESH) accredited hypertension specialist.
He is a Fellow of the American Heart Association (FAHA), the
European Society of Cardiology (FESC), and the International
Society of Hypertension (ISHF). He served as an Executive Committee
of the ISH from 2018-2020 and is currently on the Management Board
of the global ISH May Measurement Month campaign. Markus is
President of Hypertension Australia and a Trustee of the Foundation
for High Blood Pressure Research.
Markus has a strong background in clinical research with a focus
on the pathophysiology of hypertension, involvement of the kidneys,
and hypertension mediated organ damage. He has a specific interest
in treatment modalities targeting the sympathetic nervous system
and other relevant pathways such as the endothelin system to
improve BP control and thereby outcomes for patients with difficult
to control hypertension. For his work he received the Björn Folkow
Award from the European Society of Hypertension (ESH) and the
Arthur C. Corcoran Award from the AHA Hypertension Council, both in
2021. He has authored more than 450 articles in peer-reviewed
journals and serves on the Editorial Board of Hypertension and
Journal of Hypertension. Prof. Schlaich serves as a consultant to
Idorsia.
About IdorsiaIdorsia Ltd is reaching out for
more – We have more ideas, we see more opportunities and we want to
help more patients. In order to achieve this, we will develop
Idorsia into a leading biopharmaceutical company, with a strong
scientific core.
Headquartered near Basel, Switzerland – a European biotech-hub –
Idorsia is specialized in the discovery, development and
commercialization of small molecules to transform the horizon of
therapeutic options. Idorsia has a 20-year heritage of drug
discovery, a broad portfolio of innovative drugs in the pipeline,
an experienced team of professionals covering all disciplines from
bench to bedside, and commercial operations in Europe, Japan, and
the US – the ideal constellation for bringing innovative medicines
to patients.
Idorsia was listed on the SIX Swiss Exchange (ticker symbol:
IDIA) in June 2017 and has over 1,300 highly qualified specialists
dedicated to realizing our ambitious targets.
For further information, please
contactAndrew C. WeissSenior Vice President, Head
of Investor Relations & Corporate CommunicationsIdorsia
Pharmaceuticals Ltd, Hegenheimermattweg 91, CH-4123 Allschwil+41 58
844 10 10investor.relations@idorsia.commedia.relations@idorsia.com
www.idorsia.com
The above information contains certain "forward-looking
statements", relating to the company's business, which can be
identified by the use of forward-looking terminology such as
"estimates", "believes", "expects", "may", "are expected to",
"will", "will continue", "should", "would be", "seeks", "pending"
or "anticipates" or similar expressions, or by discussions of
strategy, plans or intentions. Such statements include descriptions
of the company's investment and research and development programs
and anticipated expenditures in connection therewith, descriptions
of new products expected to be introduced by the company and
anticipated customer demand for such products and products in the
company's existing portfolio. Such statements reflect the current
views of the company with respect to future events and are subject
to certain risks, uncertainties and assumptions. Many factors could
cause the actual results, performance or achievements of the
company to be materially different from any future results,
performances or achievements that may be expressed or implied by
such forward-looking statements. Should one or more of these risks
or uncertainties materialize, or should underlying assumptions
prove incorrect, actual results may vary materially from those
described herein as anticipated, believed, estimated or
expected.
Idorsia (LSE:0RQE)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
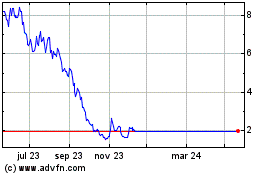
Idorsia (LSE:0RQE)
Gráfica de Acción Histórica
De May 2023 a May 2024