ImmuPharma Shares Rise on Positive Guidance From US FDA for P140
18 Mayo 2023 - 4:38AM
Noticias Dow Jones
By Kyle Morris
Shares in ImmuPharma rose 11% on Thursday after the company said
it received positive guidance from the U.S. Food and Drug
Administration that supports an Investigational New Drug
application and a phase 2/3 adaptive clinical trial of the P140
platform for patients with chronic idiopathic demyelinating
polyneuropathy.
Shares at 0850 GMT were up 0.33 pence at 3.40 pence.
The company said the FDA feedback recognised that P140 is
suitable to be studied in another disease indication in addition to
systemic lupus erythematosus, which supports P140 across several
auto-immune/inflammatory diseases.
An IND application will now be prepared for submission to the
FDA.
An application for Orphan Drug status for CIDP will be also
submitted in parallel to the full IND application.
Write to Kyle Morris at kyle.morris@dowjones.com
(END) Dow Jones Newswires
May 18, 2023 05:23 ET (09:23 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
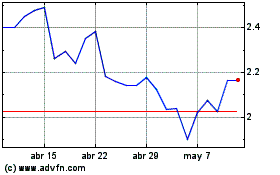
Immupharma (LSE:IMM)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
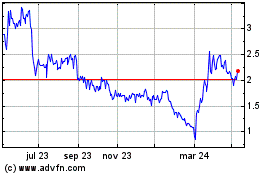
Immupharma (LSE:IMM)
Gráfica de Acción Histórica
De May 2023 a May 2024