Impel Pharmaceuticals to Present Data on Trudhesa® Nasal Spray for Treatment of Acute Migraine at 2023 American Academy of Neurology Annual Meeting
11 Abril 2023 - 7:00AM

Impel Pharmaceuticals (NASDAQ: IMPL), a commercial-stage
pharmaceutical company developing transformative therapies for
people suffering from diseases with high unmet medical needs,
announced today that four Trudhesa® (dihydroergotamine mesylate)
nasal spray (0.725 mg per spray) abstracts have been selected to be
presented at the upcoming American Academy of Neurology (AAN)
Annual Meeting taking place in Boston, MA, April 22-27, 2023.
The Trudhesa posters to be presented are as
follows:
- A Cross-Sectional Survey of Prevailing Opinions from
Headache Specialists Regarding Status Migrainosus
Management; (Poster 12-001; Session 11)
- Presentation Time: Wednesday, April 26, 2023, 11:45 a.m.
ET
- Clinical Collaborators: Jennifer Robblee, M.D., Barrow
Neurological Institute, Phoenix, AZ
- Assessment of the Potential for Drug–Drug Interactions
Between INP104 and Gepants for Migraine Management Using a
Model-based Approach (Poster 12-008; Session 12)
- Clinical Collaborators: Sutapa Ray, PhD., Impel
Pharmaceuticals, Seattle, WA
- Presentation Time: Wednesday, April 26, 2023, 5:30 p.m. ET
- Safety and Efficacy of Concomitant Erenumab and INP104
Use from the Phase 3 STOP 301 Study in Migraine Patients
(Poster 12-002; Session 13)
- Presentation Time: Thursday, April 27, 2023, 8:00 a.m. ET
- Clinical Collaborators: Zubair Ahmed, M.D., Cleveland Clinic,
Cleveland, OH
- Safety of Concomitant Triptan and INP104 Use from the
Phase 3 STOP 301 Study in Migraine Patients (Poster
12-011; Session 14)
- Presentation Time: Thursday, April 27, 2023, 11:45 a.m. ET
- Clinical Collaborators: Alexander Feoktistov, M.D., Synergy
Integrative Headache Center, Northfield, IL
The full abstracts can be found on the AAN
website.
About Impel
PharmaceuticalsImpel Pharmaceuticals is a commercial-stage
pharmaceutical company developing transformative therapies for
people suffering from diseases with high unmet medical needs. Impel
offers treatments that pair its proprietary POD® technology
with well-established therapeutics. In September 2021, Impel
received U.S. FDA approval for its first product,
Trudhesa® nasal spray, which is approved in the U.S. for the
acute treatment of migraine with or without aura in adults. In
addition to Trudhesa, the Company continues to address patient
needs via licensing and partnerships.
About Impel’s Precision Olfactory
Delivery (POD®) Technology:Impel’s proprietary POD®
technology is able to deliver a range of therapeutic molecules and
formulations into the vascular-rich upper nasal space, believed to
be a gateway for unlocking the previously unrealized full potential
of these molecules. By delivering predictable doses of drug
directly to the upper nasal space, Impel’s precision performance
technology has the goal of enabling increased and consistent
absorption of drug, overriding the high variability associated with
other nasal delivery systems, yet without the need for an
injection. While an ideal target for drug administration, to date
no technology has been able to consistently deliver drugs to the
upper nasal space. By utilizing this route of administration, Impel
Pharmaceuticals has been able to demonstrate blood concentration
levels for its investigational therapies that are comparable to
intramuscular (IM) administration and can even reach intravenous
(IV)-like systemic levels quickly. Importantly, the POD technology
offers propellant-enabled delivery of dry powder and liquid
formulations that eliminates the need for coordination of
breathing, allowing for self- or caregiver-administration in a
manner that may improve patient outcome, comfort, and potentially,
compliance.
About Trudhesa®
IndicationTrudhesa® is used to treat an active
migraine headache with or without aura in adults. Do not use
Trudhesa to prevent migraine when you have no symptoms. It is not
known if Trudhesa is safe and effective in children.
Important Safety
Information
|
Serious or potentially life-threatening reductions in blood flow to
the brain or extremities due to interactions between
dihydroergotamine (the active ingredient in Trudhesa) and strong
CYP3A4 inhibitors (such as protease inhibitors and macrolide
antibiotics) have been reported rarely. As a result, these
medications should not be taken together. |
Do not use Trudhesa if you:
- Have any disease affecting your
heart, arteries, or blood circulation.
- Are taking certain anti-HIV
medications known as protease inhibitors (such as ritonavir or
nelfinavir).
- Are taking a macrolide antibiotic
such as clarithromycin or erythromycin.
- Are taking certain antifungals such
as ketoconazole or itraconazole.
- Have taken certain medications such
as triptans or ergot-type medications for the treatment or
prevention of migraine within the last 24 hours.
- Have taken any medications that
constrict your blood vessels or raise your blood pressure.
- Have severe liver or kidney
disease.
- Are allergic to ergotamine or
dihydroergotamine.
Before taking Trudhesa, tell your doctor
if:
- You have high blood pressure, chest
pain, shortness of breath, heart disease; or risk factors for heart
disease (such as high blood pressure, high cholesterol, obesity,
diabetes, smoking, strong family history of heart disease or you
are postmenopausal, or male over 40); or problems with blood
circulation in your arms, legs, fingers, or toes.
- You have or had any disease of the
liver or kidney.
- You are taking any prescription or
over-the-counter medications, including vitamins or herbal
supplements.
- You are pregnant, planning to
become pregnant or are nursing, or have ever stopped medication due
to an allergy or bad reaction.
- This headache is different from
your usual migraine attacks.
The use of Trudhesa should not exceed dosing
guidelines and should not be used on a daily basis. Serious cardiac
(heart) events, including some that have been fatal, have occurred
following the use of dihydroergotamine mesylate, particularly with
dihydroergotamine for injection, but are extremely rare.
You may experience some nasal congestion or
irritation, altered sense of taste, sore throat, nausea, vomiting,
dizziness, and fatigue after using Trudhesa. Contact your doctor
immediately if you experience:
- Numbness or tingling in your
fingers and toes
- Severe tightness, pain, pressure,
heaviness, or discomfort in your chest
- Muscle pain or cramps in your arms
or legs
- Cold feeling or color changes in
one or both legs or feet
- Sudden weakness
- Slurred speech
- Swelling or itching
The risk information provided here is not
comprehensive. To learn more, talk about Trudhesa with your
healthcare provider or pharmacist. The FDA-approved product
labeling can be found at www.Trudhesa.com or 1-800-555-DRUG. You
can also call 1-833-TRUDHESA (1-833-878-3437) for additional
information.
Cautionary Note on Forward-Looking
StatementsThis press release contains “forward-looking”
statements within the meaning of the safe harbor provisions of the
U.S. Private Securities Litigation Reform Act of 1995, including,
but not limited to, the potential clinical benefits of Trudhesa®,
the market opportunities of Trudhesa within the migraine market,
the speed of uptake and market growth of Trudhesa, the
effectiveness of the Trudhesa sales force, the timing of
announcements of clinical results and clinical development
activities of Impel’s product candidates, and Impel’s cash runway.
Forward-looking statements can be identified by words such as:
“believe,” “may,” “will,” “potentially,” “estimate,” “continue,”
“anticipate,” “intend,” “could,” “would,” “project,” “plan,”
“expect” or the negative or plural of these words or similar
expressions. These statements are subject to numerous risks and
uncertainties that could cause actual results and events to differ
materially from those anticipated, including but not limited to,
Impel’s ability to maintain regulatory approval of Trudhesa, its
ability to execute its commercialization strategy for Trudhesa, its
ability to develop, manufacture and commercialize its other product
candidates including plans for future development of its POD®
devices and plans to address additional indications for which Impel
may pursue regulatory approval, whether results of preclinical
studies or clinical trials will be indicative of the results of
future trials, and the effects of COVID-19 on its clinical programs
and business operations. Many of these risks are described in
greater detail in Impel’s filings with the Securities and Exchange
Commission. Any forward-looking statements in this press release
speak only as of the date of this press release. Impel assumes no
obligation to update forward-looking statements whether as a result
of new information, future events or otherwise, after the date of
this press release.
Impel, POD, Trudhesa and the Impel logo are
registered trademarks of Impel Pharmaceuticals Inc. To learn more
about Impel Pharmaceuticals, please visit our website at
https://impelpharma.com.
Contact:Melyssa WeibleElixir
Health Public RelationsPhone: (1)
201-723-5805Email: mweible@elixirhealthpr.com
Impel Pharmaceuticals (NASDAQ:IMPL)
Gráfica de Acción Histórica
De May 2024 a Jun 2024
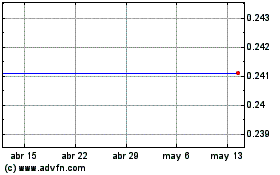
Impel Pharmaceuticals (NASDAQ:IMPL)
Gráfica de Acción Histórica
De Jun 2023 a Jun 2024