Crossject appoints Tony Tipton as U.S. Chief Operating Officer in Preparation for Commercialization of ZEPIZURE® Epilepsy Rescue Treatment
19 Agosto 2024 - 10:30AM

Press Release
Crossject appoints Tony Tipton as U.S.
Chief Operating Officer in Preparation for Commercialization of
ZEPIZURE® Epilepsy Rescue Treatment
- Experienced executive to lead
preparations for U.S. commercialization of ZEPIZURE®
- Reinforces and de-risks
pre-commercial activities ahead of submitting marketing
authorization application for ZEPIZURE® epilepsy rescue treatment
in the U.S.
Dijon, France August 19, 2024 17:30
CET-- Crossject (ISIN: FR0011716265; Euronext: ALCJ), a specialty
pharma company developing medicines for emergency
situations harnessing its award-winning needle-free auto-injector
ZENEO®, announces the appointment of the deeply experienced
healthcare executive Tony Tipton as U.S. Chief Operating
Officer.
Mr. Tipton has over 25 years of experience
across the key pillars of commercial pharmaceuticals business,
encompassing corporate leadership, business development, market
access, sales force leadership, marketing and trade operations. He
joins Crossject from his role as Chief Operating & Commercial
Officer at Xequel Bio, where he led commercialization strategy,
pre-commercial activities for BARDA/NIH funded assets, and
commercial assets through acquisition. Mr. Tipton’s appointment
will strengthen Crossject’s U.S. pre-commercial activities as the
company prepares the ZEPIZURE® U.S. marketing authorization
application submission.
Mr. Tipton previously served in several senior
commercial roles in large international specialty pharmaceutical
companies, including Interim Chief Commercial Officer and Vice
President, Market Access & Trade Channels at Santen
Pharmaceuticals, via the acquisition of Eyevance Pharmaceuticals.
In this role, Mr. Tipton led US commercial functions, achieving
over $70 million in gross revenue for the US commercial business.
He has also held a Director’s position for Health Systems Accounts
for Sunovion Pharmaceuticals, a role in which he launched a new
epilepsy drug, Aptiom, and several sales and marketing positions at
Galderma Laboratories.
“Tony brings to Crossject a proven track record
of excellence and highly relevant experience across all facets of
precommercial, sales and marketing in the specialty pharmaceutical
industry and, specifically, for emergency medicines comparable to
ZEPIZURE®. His leadership, knowledge and experience are a perfect
fit and further inform and de-risk our move towards
commercialization of ZEPIZURE® in the US, our key market. We look
forward to the dynamic contributions he will bring to our team,”
said Patrick Alexandre, CEO of Crossject.
“My senior leadership roles in preparing
companies for commercialization have provided me with relevant
experience, valuable networks and strategic insights, all of which
I leverage in my role at Crossject. Joining Crossject at this
pivotal time is a responsibility that I am ready to embrace with
energy and enthusiasm as we bring lifesaving, innovative rescue
medicines like ZEPIZURE® to market. The ZENEO® technology platform
and Crossject’s exciting pipeline offer multiple rapid expansion
opportunities. I look forward to leveraging my background in
commercial strategies and partnering with the talented team at
Crossject as we focus on bringing the company’s first rescue
therapy to market,” commented Tony Tipton, COO of Crossject
U.S.
About Crossject
Crossject SA (Euronext:
ALCJ; www.crossject.com) is an emerging specialty
pharmaceutical company developing medicines for emergency
situations harnessing its award-winning needle-free auto-injector
ZENEO® platform. Crossject is in advanced regulatory development
for ZEPIZURE®, an epileptic rescue therapy, for which it has a $60
million contract* with the U.S. Biomedical Advanced Research and
Development Authority (BARDA). The Company’s versatile ZENEO®
platform is designed to enable patients or untrained caregivers to
easily and instantly deliver a broad range of emergency drugs via
intramuscular injection on bare skin or even through clothing. The
Company’s other products in development include mainly solutions
for allergic shocks and adrenal insufficiencies, as well as
therapies and other emergency indications.
* Contract no: 75A50122C00031 with the
Department of Health and Human Services; Administration for
Strategic Preparedness and Response; Biomedical Research and
Development Authority
For further information, please contact:
|
Investors Natasha DrapeauCohesion Bureau+41 76 823
75 27natasha.drapeau@cohesionbureau.com |
MediaSophie BaumontCohesion Bureau+33 6 27 74 74
49sophie.baumont@cohesionbureau.com |
|
- PR_CrossJect Appoints US COO_EN
Crossject (EU:ALCJ)
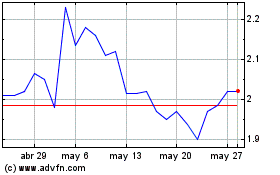
Gráfica de Acción Histórica
De Sep 2024 a Oct 2024
Crossject (EU:ALCJ)
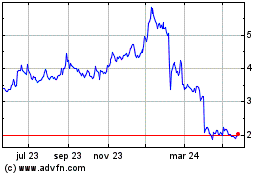
Gráfica de Acción Histórica
De Oct 2023 a Oct 2024