Atara Biotherapeutics to Submit New FDA Biologics License Application in 2Q
19 Septiembre 2023 - 3:50PM
Noticias Dow Jones
By Sabela Ojea
Atara Biotherapeutics said it plans to submit a biologics
license application for tabelecleucel following productive
conversations with the U.S. Food and Drug Administration.
The biopharmaceutical company and the FDA are now aligned and it
expects to submit the application in the second quarter of 2024,
Atara Biotherapeutics said Tuesday.
The BLA submission for tabelecleucel "will enable Atara to
incorporate the latest tab-cel pivotal trial data from the Allele
study into the BLA filing package," it said.
Tabelecleucel, approved in the European Union, is being
developed for relapsed or refractory Epstein-Barr virus-positive
post-transplant lymphoproliferative disease, a rare hematologic
cancer.
Write to Sabela Ojea at sabela.ojea@wsj.com; @sabelaojeaguix
(END) Dow Jones Newswires
September 19, 2023 16:35 ET (20:35 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
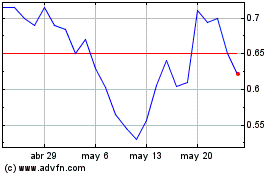
Atara Biotherapeutics (NASDAQ:ATRA)
Gráfica de Acción Histórica
De May 2024 a Jun 2024
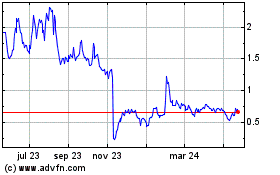
Atara Biotherapeutics (NASDAQ:ATRA)
Gráfica de Acción Histórica
De Jun 2023 a Jun 2024