0001401914
false
Dare Bioscience, Inc.
0001401914
2023-08-10
2023-08-10
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date
of earliest event reported): August 10, 2023
DARÉ BIOSCIENCE,
INC.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-36395 |
|
20-4139823 |
(State or
other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(I.R.S.
Employer
Identification No.) |
3655 Nobel Drive,
Suite 260
San Diego, CA 92122
(Address of Principal Executive Offices and Zip Code)
Registrant’s telephone
number, including area code: (858) 926-7655
Not Applicable
(Former name or former address, if changed since last report.)
Check the appropriate
box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following
provisions (see General Instruction A.2. below):
| ☐ |
Written communications pursuant
to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant
to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications
pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications
pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered
pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol(s) |
|
Name
of each exchange on which registered |
| Common
stock |
|
DARE |
|
Nasdaq
Capital Market |
Indicate by check mark
whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter)
or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company ☐
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 2.02 |
Results
of Operations and Financial Condition. |
On August 10, 2023,
Daré Bioscience, Inc. (the “Company”), issued a press release announcing its financial results for the quarter ended
June 30, 2023, a copy of which is furnished as Exhibit 99.1 to this report.
The information under
this Item 2.02 and in Exhibit 99.1 is being furnished and is not being filed for purposes of Section 18 of the Securities and Exchange
Act of 1934 and is not to be incorporated by reference into any filing of the Company under the Securities Act of 1933 or the Securities
Exchange Act of 1934, whether made before or after the date hereof, regardless of any general incorporation language in any such filing,
except as shall be expressly set forth by specific reference in such a filing.
| Item 4.01 |
Changes
in Registrant’s Certifying Accountant. |
On August 4, 2023,
Mayer Hoffman McCann P.C. (“MHM”), notified the Company that MHM decided to resign as the Company’s independent registered
public accounting firm effective upon the earlier of (i) the filing of the Company’s annual report on Form 10-K for the fiscal
year ending December 31, 2023, (ii) the date on which the Company engages a new independent registered public accounting firm and (iii)
April 1, 2024. MHM informed the Company that its decision to resign was not as a result of any action taken or not taken by the Company.
Neither the Company’s Board of Directors nor the Audit Committee of the Company’s Board of Directors were involed in
MHM’s decision to resign. The Company is in the process of selecting a new independent registered public accounting firm.
MHM audited the Company’s
consolidated financial statements for the years ended December 31, 2022 and 2021. MHM’s report dated March 30, 2023, with respect
to the consolidated financial statements of the Company as of December 31, 2022 and 2021 and for each of the years ended December 31,
2022 and December 31, 2021, did not contain an adverse opinion or a disclaimer of opinion and was not qualified or modified as to uncertainty,
audit scope, or accounting principles, except that such report contained an explanatory paragraph regarding the Company’s ability
to continue as a going concern. During the two years ended December 31, 2022, and from December 31, 2022 through August 4, 2023, there
were no (i) disagreements (as defined in Item 304(a)(1)(iv) of Regulation S-K and the related instructions to Item 304 of Regulation
S-K) between the Company and MHM on any matter of accounting principles or practices, financial statement disclosure, or auditing scope
or procedure, which disagreements, if not resolved to satisfaction of MHM, would have caused MHM to make reference to the subject matter
of such disagreements in connection with its report, or (ii) “reportable events,” as described in Item 304(a)(1)(v) of Regulation
S-K, that would require disclosure under Item 304(a)(1)(v) of Regulation S-K.
The Company furnished
MHM with a copy of the disclosure in this Item 4.01 and requested that MHM furnish the Company with a letter addressed to the U.S. Securities
and Exchange Commission stating whether MHM agrees with the statements made by the Company in this Item 4.01, and if not, stating the
respects in which it does not agree. MHM furnished such a letter, a copy of which is filed as Exhibit 16.1 to this report.
| Item 9.01 |
Financial
Statements and Exhibits. |
(d) Exhibits.
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto
duly authorized.
| |
DARÉ BIOSCIENCE,
INC. |
| |
|
| Dated:
August 10, 2023 |
By: |
/s/
Sabrina Martucci Johnson |
| |
Name: |
Sabrina
Martucci Johnson |
| |
Title: |
President
and Chief Executive Officer |
Exhibit
16.1
August
10, 2023
Securities
and Exchange Commission
100
F Street N.E.
Washington,
D.C. 20549
Commissioners:
We
have read Item 4.01 of Daré Bioscience, Inc.’s Form 8-K dated August 10, 2023, and we agree with the statements set forth
in Item 4.01, insofar as they relate to our firm. We have no basis to agree or disagree with other statements of the registrant contained
therein.
Very
truly yours,
/s/
Mayer Hoffman McCann P.C.
San
Diego, California
Exhibit
99.1

Daré
Bioscience Reports Second Quarter 2023 Financial Results and Provides a Company Update
Conference
Call and Webcast Today at 4:30 p.m. ET
Second
Half 2023 Anticipated Milestones (target indication(s)):
| |
● |
XACIATO™
First Commercial Sale |
| |
● |
Ovaprene®
(hormone-free monthly contraception) – Pivotal Phase 3 Contraceptive Efficacy Study – Patient Enrollment to Begin
4Q2023 |
| |
● |
Sildenafil
Cream, 3.6% (female sexual arousal disorder and female sexual interest/arousal disorder) - preparations for end-of-Phase 2 meeting
with FDA |
| |
● |
DARE-PDM1
(primary dysmenorrhea) – Phase 1 Clinical Study Topline Data |
| |
● |
Menopause
programs: IND related activities for DARE-HRT1 (vasomotor symptoms of menopause) and DARE-VVA1 (vulvovaginal atrophy,
a common condition in postmenopausal women, including those whose menopause resulted from receiving certain breast cancer treatments);
Phase 3 and Phase 2 clinical study initiation plans, respectively |
SAN
DIEGO August 10, 2023 (GLOBE NEWSWIRE) — Daré Bioscience, Inc. (NASDAQ: DARE), a leader in women’s health innovation,
today reported financial results for the quarter ended June 30, 2023, and provided a company update.
“We
are particularly proud of the milestone we reached in June – the announcement of the topline data from our exploratory Phase 2b
RESPOND study evaluating topical Sildenafil Cream, 3.6% as a treatment for female sexual arousal disorder. This study was the first at-home,
Phase 2 clinical study by a pharmaceutical drug development company to address a distressing condition for women that is analogous to
erectile dysfunction for men utilizing a novel topical cream formulation of sildenafil, the active ingredient in Viagra®,”
said Sabrina Martucci Johnson, President and CEO of Daré Bioscience. “The group receiving Sildenafil Cream in the RESPOND
study demonstrated improvements in arousal lubrication, orgasm and sexual desire, which persisted through the end of study assessment.
Based on data from the study, demonstrating improvement in multiple facets of female sexual dysfunction, and because there is no FDA-approved
product for the treatment of female sexual arousal disorder or female sexual interest/arousal disorder, Sildenafil Cream has the potential
to be a first-in-category product with a market opportunity comparable in size to the erectile dysfunction market.”
“We
also remain very enthusiastic about rest of 2023 given two highly anticipated milestones,” said Ms. Johnson. “First, we are
on track to begin patient enrollment in what we believe will be the single pivotal Phase 3 study for Daré’s late-stage portfolio
candidate, Ovaprene, an investigational, hormone-free, monthly intravaginal contraceptive designed to be an easy-to-use monthly option
with effectiveness approaching hormonal methods and whose U.S. commercial rights are under a license agreement with Bayer. Second, we
are looking forward to the U.S. commercial launch of XACIATOTM by our global collaboration partner, Organon. Finally, later
this year we expect to announce the topline findings from our DARE-PDM1 Phase 1 trial, a multi-center, randomized, placebo-controlled,
double-blind, 3-arm parallel group study of approximately 36 healthy, premenopausal women with primary dysmenorrhea, also referred to
as menstrual cramps and pain. Prevalence rates of dysmenorrhea vary but range from 50% to 90%. Recent market research suggests that the
global market for dysmenorrhea treatment was valued at $13 billion in 2022 and is expected to increase to $28.5 billion by 2029¹.”
1H-2023
In Review and 2H-2023 Anticipated Developments
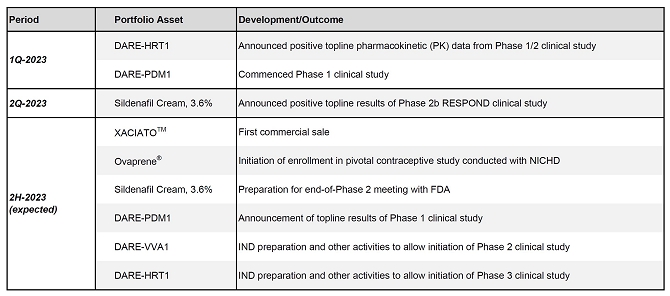
Daré
Portfolio Summary
| |
● |
One
FDA approved product, XACIATOTM |
| |
● |
13 development
programs across 9 distinct indications |
| |
● |
3 products
in, or nearing, Phase 3 clinical development |
| |
● |
Key
XACIATO, Sildenafil Cream, and Ovaprene milestones anticipated during 2H-2023 |
XACIATOTM
(clindamycin phosphate) vaginal gel, 2%
A
lincosamide antibacterial indicated for the treatment of bacterial vaginosis in female patients 12 years of age and older. Please
click here for full prescribing information.
| |
○ |
3Q-2022:
$10.0 million cash payment received under commercial license agreement with Organon |
| |
○ |
2H-2023:
$1.0 million cash received, and $1.8 million milestone anticipated upon 1st commercial sale |
| |
|
|
| |
|
Bacterial
vaginosis is the most common cause of vaginitis worldwide and is estimated to affect approximately 23 million women in the U.S.2
The condition results from an overgrowth of bacteria, which upsets the balance of the natural vaginal microbiome and can lead
to symptoms of odor and discharge. In addition to being the most common type of vaginal infection in women of reproductive age and
having bothersome symptoms, bacterial vaginosis has been associated with certain increased health risks, including pre-term labor
and infertility. 2, 3 |
Ovaprene®
A
novel, investigational hormone-free monthly intravaginal contraceptive whose U.S. commercial rights are under a license agreement with
Bayer HealthCare.
| |
○ |
4Q-2022:
Investigator meeting held (with NICHD) for the pivotal Phase 3 clinical study |
| |
○ |
4Q-2023:
Anticipated initiation of subject enrollment in the pivotal Phase 3 clinical study, a single arm, open-label contraceptive efficacy
study over 12-months of use (13 menstrual cycles) |
| |
|
|
| |
|
The
pivotal Phase 3 clinical study will be conducted under a Cooperative Research and Development Agreement with the U.S. Department
of Health and Human Services, as represented by the Eunice Kennedy Shriver National Institute of Child Health and Human Development
(NICHD), part of the National Institutes of Health. |
Sildenafil
Cream, 3.6%
A
proprietary, investigational cream formulation of sildenafil, the active ingredient in Viagra®, for topical on-demand administration
to treat female sexual arousal disorder and/or female sexual interest/arousal disorder.
| |
○ |
2Q-2023:
Initiated Phase 1 thermography study with expected completion in 2023 |
| |
○ |
2Q-2023:
Announced positive topline data from exploratory Phase 2b RESPOND clinical study |
| |
|
|
| |
|
Activities
related to psychometric analyses to further refine the measures and resulting endpoints from the Phase 2b RESPOND study for use in
a Phase 3 pivotal clinical study and preparations for data review in an end-of-Phase 2 meeting with the FDA are underway. |
DARE-HRT1
A
unique, investigational intravaginal ring designed to deliver bio-identical estradiol and progesterone continuously over a 28-day period
for the treatment of moderate to severe vasomotor symptoms, as part of menopausal hormone therapy.
| |
○ |
4Q-2022:
Positive topline efficacy data reported from Phase 1/2 clinical study |
| |
○ |
1Q-2023:
Positive topline PK data reported from Phase 1/2 clinical study, and plans to progress to a single Phase 3 study announced |
| |
○ |
2H-2023:
Activities underway to support an Investigational New Drug application (IND) submission and Phase 3 clinical study initiation |
DARE-VVA1
A
proprietary, investigational formulation of tamoxifen for intravaginal administration to treat vulvovaginal atrophy in women without
the use of hormones.
| |
○ |
4Q-2022:
Positive topline safety, tolerability, PK and pharmacodynamics data reported from Phase 1/2 clinical study |
| |
○ |
2H-2023:
Activities underway to support IND submission and Phase 2 clinical study initiation |
Financial
Highlights for the Quarter ended June 30, 2023
| |
● |
Cash
and cash equivalents: $13.3 million at June 30, 2023. |
| |
● |
General
and administrative expenses: $2.9 million in 2Q-2023 as compared to $2.8 million in 2Q-2022, with the current quarter’s increase
primarily attributable to increases in personnel costs, professional services expense, and stock-based compensation expense, partially
offset by decreases in general corporate overhead expenses. |
| |
● |
Research
and development expenses: $6.0 million in 2Q-2023 as compared to $6.8 million in 2Q-2022, with the current quarter’s decrease
primarily attributable to decreases in expenses related to our Sildenafil Cream Phase 2b RESPOND clinical study, and clinical trial
and manufacturing and regulatory activities for Ovaprene, partially offset by increases in costs related to development activities
for our Phase 1 and Phase 1-ready programs. |
1 https://www.reanin.com/report-store/healthcare/pharmaceuticals-and-therapeutics/dysmenorrhea-treatment/global-dysmenorrhea-treatment-market,
accessed 5 August 2023
2
https://www.cdc.gov/std/bv/stats.htm and https://www.census.gov/data/datasets/2017/demo/popproj/2017-popproj.html,
accessed 5 August 2023
3
https://www.mayoclinic.org/diseases-conditions/bacterial-vaginosis/symptoms-causes/syc-20352279, accessed 5 August 2023
Conference
Call
Daré
will host a conference call and live webcast today at 4:30 p.m. Eastern Time to review financial results for the quarter ended June 30,
2023 and to provide a company update.
To
access the conference call via phone, dial (800) 715-9871 (U.S.) or (646) 307-1963 (international). The conference ID number for the
call is 8451277. To listen to the event and view the presentation slides via live webcast, join from the Investors section of the Company’s
website at http://ir.darebioscience.com under “Presentations, Events & Webcasts.” Callers will be able to access
the presentation slides at the same location on the Company’s website. Please log in approximately 5-10 minutes prior to the call
to register and to download and install any necessary software. The webcast and presentation slides will be archived under “Presentations,
Events & Webcasts” in the Investors section of the Company’s website at http://ir.darebioscience.com and available
for replay until August 24, 2023.
About
Daré Bioscience
Daré
Bioscience is a biopharmaceutical company committed to advancing innovative products for women’s health. The company’s mission
is to identify, develop and bring to market a diverse portfolio of differentiated therapies that prioritize women’s health and
well-being, expand treatment options, and improve outcomes, primarily in the areas of contraception, vaginal health, reproductive health,
menopause, sexual health and fertility.
Daré’s
first FDA-approved product, XACIATO™ (clindamycin phosphate) vaginal gel, 2% is a lincosamide antibacterial indicated for the treatment
of bacterial vaginosis in female patients 12 years of age and older, which is under a global license agreement with Organon. Daré’s
portfolio also includes potential first-in-category candidates in clinical development: Ovaprene®, a novel, hormone-free monthly
intravaginal contraceptive whose U.S. commercial rights are under a license agreement with Bayer; Sildenafil Cream, 3.6%, a novel cream
formulation of sildenafil to treat female sexual arousal disorder (FSAD) and/or female sexual interest/arousal disorder (FSIAD) utilizing
the active ingredient in Viagra®; and DARE-HRT1, a combination bio-identical estradiol and progesterone intravaginal ring for menopausal
hormone therapy. To learn more about XACIATO, Daré’s full portfolio of women’s health product candidates, and Daré’s
mission to deliver differentiated therapies for women, please visit www.darebioscience.com.
Daré
may announce material information about its finances, product and product candidates, clinical trials and other matters using the Investors
section of its website (http://ir.darebioscience.com), SEC filings, press releases, public conference calls and webcasts. Daré
will use these channels to distribute material information about the company, and may also use social media to communicate important
information about the company, its finances, product and product candidates, clinical trials and other matters. The information Daré
posts on its investor relations website or through social media channels may be deemed to be material information. Daré encourages
investors, the media, and others interested in the company to review the information Daré posts in the Investors section of its
website and to follow these Twitter accounts: @SabrinaDareCEO and @DareBioscience. Any updates to the list of social media channels the
company may use to communicate information will be posted in the Investors section of Daré’s website.
Forward-Looking
Statements
Daré
cautions you that all statements, other than statements of historical facts, contained in this press release, are forward-looking statements.
Forward-looking statements, in some cases, can be identified by terms such as “believe,” “may,” “will,”
“estimate,” “continue,” “anticipate,” “design,” “intend,” “expect,”
“could,” “plan,” “potential,” “predict,” “seek,” “should,” “would,”
“contemplate,” “project,” “target,” “objective,” or the negative version of these words
and similar expressions. In this press release, forward-looking statements include, but are not limited to, statements relating to expectations
regarding the commercial launch of XACIATO in the U.S., including the timing of the first commercial sale of XACIATO, plans and expectations
with respect to Daré’s product candidates, including anticipated timing for commencement and conduct of clinical trials
and announcement of topline results, the potential for FDA approval of a product candidate based on a single pivotal clinical study,
the expectation that a product candidate could be a first-in-category product, and the potential market size and opportunity for a product
candidate, if approved. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause Daré’s
actual results, performance or achievements to be materially different from future results, performance or achievements expressed or
implied by the forward-looking statements in this press release, including, without limitation, risk and uncertainties related to: Daré’s
ability to raise additional capital when and as needed to advance its product candidates, execute its business strategy and continue
as a going concern; Daré’s ability to develop, obtain FDA or foreign regulatory approval for, and commercialize its product
candidates and to do so on communicated timelines; failure or delay in starting, conducting and completing clinical trials of a product
candidate; Daré’s ability to design and conduct successful clinical trials, to enroll a sufficient number of patients, to
meet established clinical endpoints, to avoid undesirable side effects and other safety concerns, and to demonstrate sufficient safety
and efficacy of its product candidates; Daré’s dependence on third parties to conduct clinical trials and manufacture and
supply clinical trial material and commercial product; the risk that positive findings in early clinical and/or nonclinical studies of
a product candidate may not be predictive of success in subsequent clinical and/or nonclinical studies of that candidate; the risk that
development of a product candidate requires more clinical or nonclinical studies than Daré anticipates; the loss of, or inability
to attract, key personnel; the effects of the COVID-19 pandemic, macroeconomic conditions and geopolitical events on Daré’s
operations, financial results and condition, and ability to achieve current plans and objectives, including the potential impact of the
pandemic on Daré’s ability to timely enroll, conduct and report results of its clinical trials and on the ability of third
parties on which Daré relies to assist in the conduct of its business to fulfill their contractual obligations to Daré;
the risk that developments by competitors make Daré’s product or product candidates less competitive or obsolete; difficulties
establishing and sustaining relationships with development and/or commercial collaborators; failure of Daré’s product or
product candidates, if approved, to gain market acceptance or obtain adequate coverage or reimbursement from third-party payers; Daré’s
ability to retain its licensed rights to develop and commercialize a product or product candidate; Daré’s ability to satisfy
the monetary obligations and other requirements in connection with its exclusive, in-license agreements covering the critical patents
and related intellectual property related to its product and product candidates; Daré’s ability to adequately protect or
enforce its, or its licensor’s, intellectual property rights; the lack of patent protection for the active ingredients in certain
of Daré’s product candidates which could expose its products to competition from other formulations using the same active
ingredients; product liability claims; governmental investigations or actions relating to Daré’s product or product candidates
or the business activities of Daré, its commercial collaborators or other third parties on which Daré relies; the impact
of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health
care cost containment; cyber attacks, security breaches or similar events that compromise Daré’s technology systems or those
of third parties on which it relies and/or significantly disrupt Daré’s business; and disputes or other developments concerning
Daré’s intellectual property rights. Daré’s forward-looking statements are based upon its current expectations
and involve assumptions that may never materialize or may prove to be incorrect. All forward-looking statements are expressly qualified
in their entirety by these cautionary statements. For a detailed description of Daré’s risks and uncertainties, you are
encouraged to review its documents filed with the SEC including Daré’s recent filings on Form 8-K, Form 10-K and Form 10-Q.
You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date on which they were made.
Daré undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date
on which they were made, except as required by law.
Contacts:
Investors
on behalf of Daré Bioscience, Inc.:
Lee
Roth / Julia Weilman
Burns
McClellan
lroth@burnsmc.com
/ jweilman@burnsmc.com
646.930.4406
/ 646.732.4443
OR
Media
on behalf of Daré Bioscience, Inc.:
Jake
Robison
Evoke
Canale
jake.robison@evokegroup.com
619.849.5383
Source:
Daré Bioscience, Inc.
Daré
Bioscience, Inc. and Subsidiaries
Condensed
Consolidated Statements of Operations and Comprehensive Income (Loss)
(Unaudited)
| | |
Three
months ended June 30, | |
| | |
2023 | | |
2022 | |
| Revenue | |
| | | |
| | |
| License
fee revenue | |
$ | - | | |
$ | 10,000,000 | |
| Total
revenue | |
| - | | |
| 10,000,000 | |
| Operating
expenses | |
| | | |
| | |
| General
and administrative | |
| 2,920,672 | | |
| 2,792,894 | |
| Research
and development | |
| 6,043,684 | | |
| 6,797,784 | |
| License
fee expense | |
| 25,000 | | |
| 25,000 | |
| Total
operating expenses | |
| 8,989,356 | | |
| 9,615,678 | |
| Income
(loss) from operations | |
| (8,989,356 | ) | |
| 384,322 | |
| Other
income | |
| 227,124 | | |
| 29,676 | |
| Net
income (loss) | |
$ | (8,762,232 | ) | |
$ | 413,998 | |
| Foreign
currency translation adjustments | |
$ | (31,151 | ) | |
$ | (135,869 | ) |
| Comprehensive
income (loss) | |
$ | (8,793,383 | ) | |
$ | 278,129 | |
| | |
| | | |
| | |
| Income (loss) per common
share: | |
| | | |
| | |
| Basic | |
$ | (0.10 | ) | |
$ | 0.00 | |
| Diluted | |
$ | (0.10 | ) | |
$ | 0.00 | |
| | |
| | | |
| | |
| Weighted average number
of shares outstanding: | |
| | | |
| | |
| Basic | |
| 86,403,117 | | |
| 84,682,765 | |
| Diluted | |
| 86,403,117 | | |
| 85,369,424 | |
Daré
Bioscience, Inc. and Subsidiaries
Condensed
Consolidated Balance Sheets Data
| | |
June
30,
2023 | | |
December
31,
2022 | |
| | |
(unaudited) | | |
| |
| Cash
and cash equivalents | |
$ | 13,329,101 | | |
$ | 34,669,605 | |
| Working
capital | |
$ | (2,457,536 | ) | |
$ | 11,414,826 | |
| Total
assets | |
$ | 23,209,954 | | |
$ | 43,826,383 | |
| Total
stockholders’ equity (deficit) | |
$ | (2,719,402 | ) | |
$ | 11,112,110 | |
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Dare Bioscience (NASDAQ:DARE)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
Dare Bioscience (NASDAQ:DARE)
Gráfica de Acción Histórica
De May 2023 a May 2024