Research on ferroptosis is gaining momentum, but the
development of small molecule inhibitors faces numerous
challenges
BEIJING, Aug. 1, 2024
/PRNewswire/ -- Glutathione peroxidase 4 (GPX4) is recognized as a
critical regulator of ferroptosis, playing a significant role in
lipid and amino acid metabolism, as well as influencing cellular
aging, oncogenesis, and cell death [1]. Targeting GPX4-mediated
ferroptosis presents a promising therapeutic strategy, particularly
in the treatment of cancer [2].
Despite its potential, GPX4's flat surface presents significant
challenges, as it lacks distinct druggable pockets (Fig. 1A).
Current inhibitors of GPX4 with cellular activity typically
covalently bind to the selenocysteine residue at position 46,
leading to poor selectivity and high toxicity (Fig. 1B).
Given this background, it is crucial to explore potential
cryptic pockets or allosteric sites on GPX4 that could influence
its biological function.
AI-Powered RiDYMO Platform Identifies Novel Binding
Sites and Non-covalent Inhibitors
"Our strategy for developing GPX4 inhibitors centers on
identifying cryptic pockets that offer improved druggability,"
stated Dr. Xiaomin Zhang, Head of
Drug Discovery at DP Technology. "The company's AI for
Science-based RiDYMO platform employs molecular dynamics
simulations to reveal conformational changes within proteins,
facilitating the discovery of novel druggable pockets. The
non-covalent molecules developed using these pockets demonstrate
enzyme inhibition and cellular activity comparable to established
covalent controls, while exhibiting improved druggability. This
highlights the RiDYMO platform's significance in drug discovery,
particularly for challenging targets."
The workflow utilized by DP Technology's research team for this
project encompassed the following steps:
1. Protein Conformation
Sampling: The GPX4 protein was simulated using Reinforced
Dynamics (RiD)[4]. As illustrated in Fig. 2, RiD surpasses
traditional Molecular Dynamics (MD) by more effectively exploring
the protein's conformational space, revealing additional metastable
conformations and cryptic sites (with pink areas indicating amino
acids concealed within the protein).
2. Induction of Druggable
Pockets: After identifying hidden sites, we conducted
further exploration and induction of these pockets using organic
solvent probes. As shown in Fig. 3, these organic solvent probes
facilitated the formation of deeper small molecule binding pockets
on the previously flat surface of the protein.
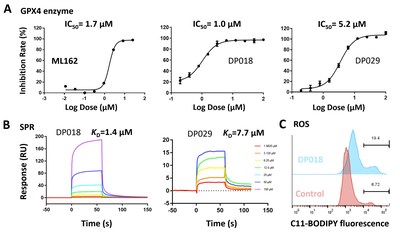
3. Molecule Screening and
Validation: High-throughput virtual screening was conducted on
these pockets using the Uni-Dock[5] from DP Technology's Hermite
drug design platform. The non-covalent molecules DP018 and DP029
exhibited micromolar-level inhibition of GPX4 enzymatic activity
(Fig. 4A), comparable to the covalent inhibitor ML162. Surface
Plasmon Resonance (SPR) experiments confirmed the direct binding of
non-covalent molecules to GPX4 with micromolar affinity (Fig.
4B). Additionally, results from the
ROS assay (Fig. 4C) indicate that these non-covalent molecules
effectively induce ferroptosis in cells.
4. Summary: James et al.
highlighted four limitations of structural biology in drug
development in their publication in Cell [6]. One of these
challenges is that protein wiggling and jiggling are crucial but
challenging to model experimentally and computationally, future
drug design will unlock methods based on the ensemble of
structures. In our research, we conducted a comprehensive analysis
of the dynamics of the protein's backbone and side chains using RiD
simulation and organic solvent probing. By effectively identifying
two druggable pockets and conducting high-throughput virtual
screening of compound libraries totaling approximately ten million
compounds, we discovered around ten active non-covalent inhibitor
molecules. Notably, two of these inhibitors demonstrated high
enzymatic activity and strong binding affinity, with their capacity
to induce ferroptosis further validated at the cellular level.
Making Proteins "Move" – The RiDYMO® Reinforced Dynamics
Platform
The RiDYMO® platform integrates various AI and physical
algorithms, dedicated to the development of drugs for "undruggable"
targets and "best-in-class" molecules. As one of its core
algorithms, Reinforced Dynamics (RiD)[4] has a significant
advantage in the sampling efficiency of molecular dynamics
simulation. By fully leveraging the high-dimensional representation
capabilities of neural networks, RiD can efficiently capture
dynamic conformational changes in complicated biomolecular systems.
Previously, the core RiD algorithm of the platform was published in
Nature Computational Science. The study demonstrated that
RiD could achieve a more comprehensive free energy surface within
1.86 μs, compared to 100 μs required by traditional MD methods,
representing nearly a hundredfold increase in efficiency.
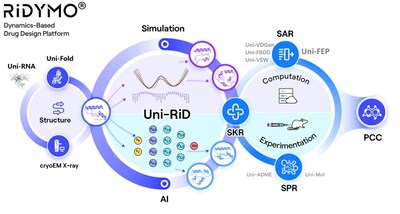
The RiDYMO® platform studies the dynamics of biological systems,
revealing cryptic binding sites across challenging systems,
including protein-protein interactions (PPIs), intrinsically
disordered proteins (IDPs), membrane proteins, and RNA. Its
effectiveness has been validated on targets such as the COPI
protein, MCL1 protein, c-Myc protein, GPX4 protein, NMDA protein,
Nav1.8 protein, and c-Myc RNA.
We look forward to collaborating with trusted partners to
advance these initiatives through all stages of development,
including hit, lead, PCC, and IND. For collaboration inquiries or
additional information, please contact bd@dp.tech.
About DP Technology
DP Technology is a global leader in the "AI for Science"
research paradigm, where AI learns scientific principles and data,
then tackles key challenges in scientific research and industrial
R&D.
We've developed the "DP Particle Universe," a suite of advanced
pre-trained models that seamlessly connect cutting-edge research
with real-world industrial applications. Our software suite
includes:
- Bohrium® Scientific Computing Space
Station
- Hermite® Computational Drug Design
Platform
- RiDYMO® Hit Discovery Platform
- Piloteye® Battery Design Automation
Platform
Together, these platforms form a robust foundation for
industrial innovation and an open ecosystem for AI in science,
fostering advancements in key areas such as drug discovery, energy,
material science, and information technology.
Relying on DP Technology's advanced Reinforced Dynamics
Platform, RiDYMO®, we have set up a world-leading hit discovery
system. The team has established external collaborations and built
up a strong internal pipeline, focusing on three areas: CNS,
oncology, and autoimmune diseases.
Reference
|
[1] Stockwell, B. R.
(2022). Ferroptosis turns 10: Emerging mechanisms, physiological
functions, and therapeutic applications. Cell,
185(14), 2401-2421.
[2] Wang, F., &
Min, J. (2021). DHODH tangoing with GPX4 on the ferroptotic stage.
Signal Transduction and Targeted Therapy, 6(1),
1-2.
[3] Moosmayer, D.,
Hilpmann, A., Hoffmann, J., Schnirch, L., Zimmermann, K., Badock,
V., ... & Hillig, R. C. (2021). Crystal structures of the
selenoprotein glutathione peroxidase 4 in its apo form and in
complex with the covalently bound inhibitor ML162. Acta
Crystallographica Section D: Structural Biology, 77(2),
237-248.
[4] Wang, D., Wang, Y.,
Chang, J., Zhang, L., Wang, H. & E, W. (2022). Efficient
sampling of high-dimensional free energy landscapes using adaptive
reinforced dynamics. Nature Computational Science,
2(1), 20-29.
[5] Yu, Y., Cai, C.,
Wang, J., Bo, Z., Zhu, Z., & Zheng, H. (2023). Uni-Dock:
GPU-accelerated docking enables ultralarge virtual screening.
Journal of chemical theory and computation, 19(11),
3336-3345.
[6] Fraser, J. S.,
& Murcko, M. A. (2024). Structure is beauty, but not always
truth. Cell, 187(3), 517-520.
|
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/novel-non-covalent-hits-against-gpx4-identified-using-the-ridymo-reinforced-dynamics-platform-of-dp-technology-302211911.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/novel-non-covalent-hits-against-gpx4-identified-using-the-ridymo-reinforced-dynamics-platform-of-dp-technology-302211911.html
SOURCE DP Technology