Ionis Pharmaceuticals, AstraZeneca Get FDA Approval for Rare Genetic Disease Treatment
21 Diciembre 2023 - 6:38PM
Noticias Dow Jones
By Ben Glickman
Ionis Pharmaceuticals and AstraZeneca's Wainua has been approved
by the U.S. Food and Drug Administration for treating a symptom of
a rare genetic disease.
The FDA approved Wainua, which has generic name eplontersen, for
the treatment of the polyneuropathy of hereditary
transthyretin-mediated amyloidosis in adults.
Ionis said Wainua would be available in the U.S. in January.
Regulatory reviews elsewhere in the world are currently
underway.
The disease, also known as hAATR, can cause polyneuropathy, a
form of nerve damage. The companies said Wainua is the only
approved medicine for treating polyneuropathy from hATTR.
The approval was based on a 35-week interim analysis from a
Phase 3 study.
Write to Ben Glickman at ben.glickman@wsj.com
(END) Dow Jones Newswires
December 21, 2023 19:23 ET (00:23 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
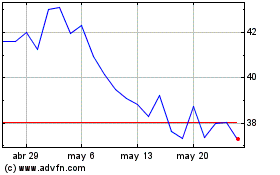
Ionis Pharmaceuticals (NASDAQ:IONS)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
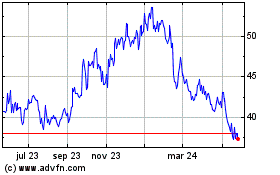
Ionis Pharmaceuticals (NASDAQ:IONS)
Gráfica de Acción Histórica
De May 2023 a May 2024