Filed Pursuant to Rule 424(b)(3)
Registration No. 333-275229
PROSPECTUS SUPPLEMENT
(to Prospectus dated November 7, 2023)

Portage Biotech Inc.
481,581 Ordinary Shares underlying Warrants
This prospectus supplement is being filed to update and supplement
the information contained in the prospectus dated November 7, 2023 (the “Prospectus”), which forms a part of our Registration
Statement on Form F-1 (Registration No. 333-275229), as amended most recently by the post-effective amendment filed on August 23, 2024,
with the information contained in our current report on Form 6-K, furnished to the Securities and Exchange Commission on November 26,
2024 (the “November 26, 2024 Form 6-K”). Accordingly, we have attached the November 26, 2024 Form 6-K to this prospectus supplement.
This prospectus supplement updates and supplements the information
in the Prospectus and is not complete without, and may not be delivered or utilized except in combination with, the Prospectus, including
any amendments or supplements thereto. This prospectus supplement should be read in conjunction with the Prospectus and if there is any
inconsistency between the information in the Prospectus and this prospectus supplement, you should rely on the information in this prospectus
supplement.
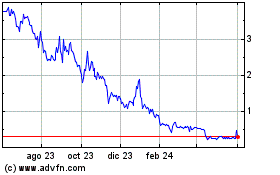
Our Ordinary Shares are listed on The Nasdaq Capital Market (“Nasdaq”)
under the symbol “PRTG”. On November 26, 2024, the closing sale price of our Ordinary Shares as reported on Nasdaq was $4.78.
Investing in the securities offered in the Prospectus involves
a high degree of risk. Before making any investment in these securities, you should consider carefully the risks and uncertainties in
the section entitled “Risk Factors” beginning on page 9 of the Prospectus, and in the other documents that are incorporated
by reference into the Prospectus.
Neither the Securities and Exchange Commission nor any state
or non-U.S. regulatory body has approved or disapproved of the securities offered in the Prospectus or passed upon the accuracy or adequacy
of the Prospectus or this prospectus supplement. Any representation to the contrary is a criminal offense.
The date of this prospectus supplement
is November 26, 2024
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of November 2024
Commission File Number: 001-40086
Portage Biotech Inc.
(Translation of registrant’s name into English)
Clarence
Thomas Building, P.O.
Box 4649, Road
Town, Tortola, British Virgin Islands, VG1110
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual
reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
INCORPORATION BY REFERENCE
This report on Form 6-K (including the exhibits attached
hereto) shall be deemed to be incorporated by reference into the registration statement on Form S-8 (File No. 333-275842) of Portage
Biotech Inc. (including any prospectuses forming a part of such registration statement) and to be a part thereof from the date on which
this report is filed, to the extent not superseded by documents or reports subsequently filed or furnished.
EXHIBITS
| Exhibit No. |
|
Exhibit |
| |
|
|
| 99.1 |
|
Unaudited Condensed Consolidated Interim Financial Statements for the three
and six months ended September 30, 2024. Unaudited - Prepared by Management as of November 26, 2024. |
| 99.2 |
|
Management’s Discussion and Analysis for the three and six months ended
September 30, 2024. |
| 101.INS |
|
Inline XBRL Instance Document. |
| 101.SCH |
|
Inline XBRL Taxonomy Extension Schema Document. |
| 101.CAL |
|
Inline XBRL Taxonomy Extension Calculation Linkbase Document |
| 101.DEF |
|
Inline XBRL Taxonomy Definition Linkbase Document |
| 101.LAB |
|
Inline XBRL Taxonomy Extension Label Linkbase Document |
| 101.PRE |
|
Inline XBRL Taxonomy Extension Presentation Linkbase Document |
| 104 |
|
Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange
Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
PORTAGE BIOTECH INC.
| By: |
/s/ Andrea Park |
|
| |
Andrea Park |
|
| |
Chief Financial Officer |
|
Exhibit 99.1
Portage Biotech Inc.
Condensed Consolidated Interim Financial Statements
For the Three and Six Months Ended September 30, 2024 and 2023
(Unaudited – Prepared by Management as of November 26,
2024)
(Expressed in U.S. Dollars)
Portage Biotech Inc.
Condensed Consolidated Interim Financial Statements
NOTICE TO READER OF CONDENSED CONSOLIDATED INTERIM FINANCIAL STATEMENTS
The condensed consolidated interim financial statements of Portage Biotech
Inc. are comprised of the condensed consolidated interim statements of financial position as of September 30, 2024 and March 31, 2024, the condensed consolidated interim statements of operations and other comprehensive
income (loss) for the three and six months ended September 30, 2024 and 2023 and the condensed consolidated interim statements of changes
in shareholders’ equity and the condensed consolidated interim statements of cash flows for the six months ended September 30, 2024
and 2023, and are the responsibility of Portage Biotech Inc.’s management.
The condensed consolidated interim financial statements of Portage Biotech
Inc. have been prepared by Portage Biotech Inc.’s management and include the selection of appropriate accounting principles, judgments
and estimates necessary to prepare these condensed consolidated interim financial statements in accordance with International Financial
Reporting Standards.
| /s/ Andrea Park |
/s/ Ian Walters |
| Andrea Park, Chief Financial Officer |
Ian Walters, MD, Chairman of the Board and Chief Executive Officer |
| |
|
| DATE: November 26, 2024 |
|
PORTAGE BIOTECH INC.
Condensed Consolidated Interim Statements of Financial Position
(U.S. Dollars in thousands)
(Unaudited – see Notice to Reader dated November 26,
2024)
| | |
| |
| | | |
| | |
| | |
Notes | |
September 30,
2024 | |
March 31,
2024 |
| | |
| |
| | | |
| | |
| Assets | |
| |
| | | |
| | |
| Current assets | |
| |
| | | |
| | |
| Cash and cash equivalents | |
14 | |
$ | 1,764 | | |
$ | 5,028 | |
| Prepaid expenses and other current assets | |
5 | |
| 922 | | |
| 2,667 | |
| Total current assets | |
| |
| 2,686 | | |
| 7,695 | |
| Non-current assets | |
| |
| | | |
| | |
| Right to use asset | |
| |
| 20 | | |
| 35 | |
| Other assets | |
| |
| - | | |
| 49 | |
| Total non-current assets | |
| |
| 20 | | |
| 84 | |
| Total assets | |
| |
$ | 2,706 | | |
$ | 7,779 | |
| | |
| |
| | | |
| | |
| Liabilities and Equity | |
| |
| | | |
| | |
| Current liabilities | |
| |
| | | |
| | |
| Accounts payable and accrued liabilities | |
7 | |
$ | 845 | | |
$ | 2,836 | |
| Lease liability - current, including interest | |
| |
| 28 | | |
| 40 | |
| Other current liabilities | |
| |
| - | | |
| 3 | |
| Total current liabilities | |
| |
| 873 | | |
| 2,879 | |
| Non-current liabilities | |
| |
| | | |
| | |
| Lease liability - non-current | |
| |
| – | | |
| 7 | |
| Warrant liability | |
6 | |
| 1,138 | | |
| 1,564 | |
| Total non-current liabilities | |
| |
| 1,138 | | |
| 1,571 | |
| Total liabilities | |
| |
| 2,011 | | |
| 4,450 | |
| | |
| |
| | | |
| | |
| Shareholders’ Equity | |
| |
| | | |
| | |
| Capital stock | |
9 | |
| 219,606 | | |
| 219,499 | |
| Stock option reserve | |
10 | |
| 24,126 | | |
| 23,841 | |
| Accumulated deficit | |
| |
| (242,334 | ) | |
| (239,318 | ) |
| Total equity attributable to owners of the Company | |
| |
| 1,398 | | |
| 4,022 | |
| Non-controlling interest | |
16 | |
| (703 | ) | |
| (693 | ) |
| Total equity | |
| |
| 695 | | |
| 3,329 | |
| Total liabilities and equity | |
| |
$ | 2,706 | | |
$ | 7,779 | |
| Commitments and Contingent Liabilities (Note 12) | |
| |
| – | | |
| | |
The accompanying notes are an integral part of these condensed consolidated
interim financial statements.
PORTAGE BIOTECH INC.
Condensed Consolidated Interim Statements of Operations and Other Comprehensive Income (Loss)
(U.S. Dollars in thousands, except per share amounts)
(Unaudited – see Notice to Reader dated November 26,
2024)
| | |
| |
| | | |
| | | |
| | | |
| | |
| | |
Note | |
Three Months Ended
September 30, | |
Six Months Ended
September 30, |
| | |
| |
2024 | |
2023 | |
2024 | |
2023 |
| Expenses | |
| |
| |
| |
| |
|
| Research and development | |
| |
$ | 723 | | |
$ | 4,237 | | |
$ | 2,028 | | |
$ | 7,865 | |
| General and administrative expenses | |
| |
| 881 | | |
| 1,693 | | |
| 2,415 | | |
| 3,062 | |
| Loss from operations | |
| |
| (1,604 | ) | |
| (5,930 | ) | |
| (4,443 | ) | |
| (10,927 | ) |
| Change in fair value of warrant liability | |
6 | |
| (716 | ) | |
| – | | |
| 426 | | |
| – | |
| Change in fair value of deferred purchase price payable – Tarus and deferred obligation – iOx milestone | |
| |
| – | | |
| (113 | ) | |
| – | | |
| (1,224 | ) |
| Gain on settlement with Parexel – iOx CRO | |
12 | |
| 946 | | |
| – | | |
| 946 | | |
| – | |
| Share of loss in associate accounted for using equity method | |
| |
| – | | |
| (40 | ) | |
| – | | |
| (90 | ) |
| Depreciation expense | |
| |
| (7 | ) | |
| (15 | ) | |
| (15 | ) | |
| (26 | ) |
| Foreign exchange transaction (loss) gain | |
| |
| (5 | ) | |
| (17 | ) | |
| (7 | ) | |
| 1 | |
| Interest income, net | |
| |
| 24 | | |
| 43 | | |
| 69 | | |
| 123 | |
| Loss before benefit for income taxes | |
| |
| (1,362 | ) | |
| (6,072 | ) | |
| (3,024 | ) | |
| (12,143 | ) |
| Income tax benefit (expense) | |
8 | |
| - | | |
| 907 | | |
| (2 | ) | |
| 1,052 | |
| Net loss | |
| |
| (1,362 | ) | |
| (5,165 | ) | |
| (3,026 | ) | |
| (11,091 | ) |
| Other comprehensive income (loss) | |
| |
| | | |
| | | |
| | | |
| | |
| Net unrealized (loss) gain on investments | |
| |
| – | | |
| (1,300 | ) | |
| – | | |
| 469 | |
| Total comprehensive loss for period | |
| |
$ | (1,362 | ) | |
$ | (6,465 | ) | |
$ | (3,026 | ) | |
$ | (10,622 | ) |
| | |
| |
| | | |
| | | |
| | | |
| | |
| Net loss attributable to: | |
| |
| | | |
| | | |
| | | |
| | |
| Owners of the Company | |
| |
$ | (1,360 | ) | |
$ | (5,158 | ) | |
$ | (3,016 | ) | |
$ | (11,077 | ) |
| Non-controlling interest | |
16 | |
| (2 | ) | |
| (7 | ) | |
| (10 | ) | |
| (14 | ) |
| Net loss | |
| |
$ | (1,362 | ) | |
$ | (5,165 | ) | |
$ | (3,026 | ) | |
$ | (11,091 | ) |
| | |
| |
| | | |
| | | |
| | | |
| | |
| Comprehensive loss attributable to: | |
| |
| | | |
| | | |
| | | |
| | |
| Owners of the Company | |
| |
$ | (1,360 | ) | |
$ | (6,458 | ) | |
$ | (3,016 | ) | |
$ | (10,608 | ) |
| Non-controlling interest | |
16 | |
| (2 | ) | |
| (7 | ) | |
| (10 | ) | |
| (14 | ) |
| Total comprehensive loss for period | |
| |
$ | (1,362 | ) | |
$ | (6,465 | ) | |
$ | (3,026 | ) | |
$ | (10,622 | ) |
| | |
| |
| | | |
| | | |
| | | |
| | |
| Loss per share | |
11 | |
| | | |
| | | |
| | | |
| | |
| Basic and diluted | |
| |
$ | (1.26 | ) | |
$ | (5.80 | ) | |
$ | (2.84 | ) | |
$ | (12.47 | ) |
| | |
| |
| | | |
| | | |
| | | |
| | |
| Weighted average shares outstanding | |
11 | |
| | | |
| | | |
| | | |
| | |
| Basic and diluted | |
| |
| 1,076 | | |
| 890 | | |
| 1,063 | | |
| 888 | |
The accompanying notes are an integral part of these condensed consolidated
interim financial statements.
PORTAGE BIOTECH INC.
Condensed Consolidated Interim Statements of Changes in Shareholders’ Equity
For the Six Months Ended September 30, 2024 and 2023
(Unaudited – see Notice to Reader dated November 26,
2024)
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| |
Accumulated | |
| |
Equity | |
| |
|
| | |
Number | |
| |
Stock | |
Other | |
| |
Attributable | |
Non- | |
|
| | |
of | |
Capital | |
Option | |
Comprehensive | |
(Accumulated | |
to Owners | |
Controlling | |
Total |
| | |
Shares | |
Stock | |
Reserve | |
Loss | |
Deficit) | |
of Company | |
Interest | |
Equity |
| | |
| In 000’ | | |
| In 000’$ | | |
| In 000’$ | | |
| In 000’$ | | |
| In 000’$ | | |
| In 000’$ | | |
| In 000’$ | | |
| In 000’$ | |
| Balance, April 1, 2024 (Pre-Split) | |
| 19,784 | | |
| | | |
| – | | |
| – | | |
| – | | |
| – | | |
| – | | |
| | |
| Balance, April 1, 2024 (After 1-for-20 reverse stock split) | |
| 989 | | |
$ | 219,499 | | |
$ | 23,841 | | |
$ | – | | |
$ | (239,318 | ) | |
$ | 4,022 | | |
$ | (693 | ) | |
$ | 3,329 | |
| Share-based compensation expense | |
| – | | |
| – | | |
| 285 | | |
| – | | |
| – | | |
| 285 | | |
| – | | |
| 285 | |
| Pre-Funded warrants exercised | |
| 60 | | |
| 1 | | |
| – | | |
| – | | |
| – | | |
| 1 | | |
| – | | |
| 1 | |
| Additional round up shares issued for fractional shares in connection with 1-for-20 reverse stock split | |
| 52 | | |
| – | | |
| – | | |
| – | | |
| – | | |
| – | | |
| – | | |
| – | |
| Shares issued for accrued bonuses | |
| 14 | | |
| 100 | | |
| - | | |
| – | | |
| – | | |
| 100 | | |
| – | | |
| 100 | |
| Shares issued for restricted
stock units, net | |
| 1 | | |
| 6 | | |
| - | | |
| – | | |
| – | | |
| 6 | | |
| – | | |
| 6 | |
| Net loss for period | |
| – | | |
| – | | |
| – | | |
| – | | |
| (3,016 | ) | |
| (3,016 | ) | |
| (10 | ) | |
| (3,026 | ) |
| Balance, September 30, 2024 | |
| 1,116 | | |
$ | 219,606 | | |
$ | 24,126 | | |
$ | – | | |
$ | (242,334 | ) | |
$ | 1,398 | | |
$ | (703 | ) | |
$ | 695 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance, April 1, 2023 (Pre-Split) | |
| 17,606 | | |
| | | |
| – | | |
| – | | |
| – | | |
| – | | |
| – | | |
| | |
| Balance, April 1, 2023 (After 1-for-20 reverse stock split) | |
| 880 | | |
$ | 218,782 | | |
$ | 21,204 | | |
$ | (4,325 | ) | |
$ | (159,616 | ) | |
$ | 76,045 | | |
$ | (650 | ) | |
$ | 75,395 | |
| Share-based compensation expense | |
| – | | |
| – | | |
| 1,512 | | |
| – | | |
| – | | |
| 1,512 | | |
| – | | |
| 1,512 | |
| Shares issued under ATM | |
| 9 | | |
| 682 | | |
| – | | |
| – | | |
| – | | |
| 682 | | |
| – | | |
| 682 | |
| Share issuance costs | |
| – | | |
| (20 | ) | |
| – | | |
| – | | |
| – | | |
| (20 | ) | |
| – | | |
| (20 | ) |
| Shares issued or accrued for services | |
| 1 | | |
| 50 | | |
| – | | |
| – | | |
| – | | |
| 50 | | |
| – | | |
| 50 | |
| Net unrealized gain on investments | |
| – | | |
| – | | |
| – | | |
| 469 | | |
| – | | |
| 469 | | |
| – | | |
| 469 | |
| Net loss for period | |
| – | | |
| – | | |
| – | | |
| – | | |
| (11,077 | ) | |
| (11,077 | ) | |
| (14 | ) | |
| (11,091 | ) |
| Balance, September 30, 2023 | |
| 890 | | |
$ | 219,494 | | |
$ | 22,716 | | |
$ | (3,856 | ) | |
$ | (170,693 | ) | |
$ | 67,661 | | |
$ | (664 | ) | |
$ | 66,997 | |
The accompanying notes are an integral part of these condensed consolidated
interim financial statements.
PORTAGE BIOTECH INC.
Condensed Consolidated Interim Statements of Cash Flows
For the Six Months Ended September 30, 2024 and 2023
(U.S. Dollars in thousands)
(Unaudited – see Notice to Reader dated November 26,
2024)
| | |
| | | |
| | |
| | |
Six Months Ended September 30, |
| | |
2024 | |
2023 |
| Cash flows from operating activities: | |
| | | |
| | |
| Net loss for the period | |
$ | (3,026 | ) | |
$ | (11,091 | ) |
| Adjustments for non-cash items: | |
| | | |
| | |
| Share-based compensation expense | |
| 285 | | |
| 1,512 | |
| Change in fair value of warrant liability | |
| (426 | ) | |
| – | |
| Gain on settlement with Parexel – iOx CRO | |
| (946 | ) | |
| – | |
| Change in fair value of deferred purchase price payable – Tarus and deferred obligation – iOx milestone | |
| – | | |
| 1,224 | |
| Decrease in deferred tax liability | |
| – | | |
| (1,063 | ) |
| Share of loss in associate | |
| – | | |
| 90 | |
| Fair value of shares issued for services | |
| – | | |
| 50 | |
| Depreciation | |
| 15 | | |
| 26 | |
| Changes in operating working capital: | |
| | | |
| | |
| Prepaid expenses and other receivables | |
| 1,216 | | |
| 286 | |
| Other assets | |
| 38 | | |
| (3 | ) |
| Accounts payable and accrued liabilities | |
| (399 | ) | |
| 1,224 | |
| Other | |
| (3 | ) | |
| – | |
| Net cash used in operating activities | |
| (3,246 | ) | |
| (7,745 | ) |
| | |
| | | |
| | |
| Cash flows from financing activities: | |
| | | |
| | |
| Proceeds from the exercise of Pre-Funded Warrants | |
| 1 | | |
| – | |
| Proceeds from shares issued under ATM and Committed Purchase Agreement | |
| – | | |
| 682 | |
| Share issuance costs | |
| – | | |
| (20 | ) |
| Repayment of lease liability | |
| (19 | ) | |
| (17 | ) |
| Net cash (used in) provided by financing activities | |
| (18 | ) | |
| 645 | |
| | |
| | | |
| | |
| Decrease in cash and cash equivalents during the period | |
| (3,264 | ) | |
| (7,100 | ) |
| Cash and cash equivalents at beginning of the period | |
| 5,028 | | |
| 10,545 | |
| Cash and cash equivalents at end of the period | |
$ | 1,764 | | |
$ | 3,445 | |
| | |
| | | |
| | |
| Supplemental disclosure of cash flow information: | |
| | | |
| | |
| Cash paid for interest | |
$ | 3 | | |
$ | 13 | |
| | |
| | | |
| | |
| Supplemental disclosure of non-cash investing and financing activities: | |
| | | |
| | |
| Right to use asset acquired | |
$ | – | | |
$ | 303 | |
| Lease liability incurred | |
$ | – | | |
$ | 303 | |
The accompanying notes are an integral part of these condensed consolidated
interim financial statements.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 1. NATURE OF OPERATIONS
Portage Biotech Inc. (the “Company” or “Portage”)
is incorporated in the British Virgin Islands (“BVI”) with its registered office located at Clarence Thomas Building, P.O.
Box 4649, Road Town, Tortola, BVI. Its USA agent, Portage Development Services Inc. (“PDS”), is located at 59 Wilton Road,
Westport, CT, 06880, USA.
The Company is a foreign private issuer under the Securities
and Exchange Commission (the “SEC”) rules. It is also a reporting issuer under the securities legislation of the provinces
of Ontario and British Columbia. Its ordinary shares were listed on the Canadian Securities Exchange (“CSE”) under the symbol
“PBT.U”. On February 25, 2021, the ordinary shares of the Company began trading on the Nasdaq Capital Market (“Nasdaq”)
under the symbol “PRTG”. As the principal market for the Company’s ordinary shares is Nasdaq, the Company voluntarily
delisted from the CSE on April 23, 2021.
Portage is a clinical-stage immuno-oncology company advancing
treatments the Company believes will be first-in class therapies that target known checkpoint resistance pathways to improve long-term
treatment response and quality of life in patients with invasive cancers. Portage’s access to next-generation technologies coupled
with a deep understanding of biological mechanisms enables the identification of clinical therapies and product development strategies
that accelerate these medicines through the translational pipeline. After a review of the Company’s future funding needs for clinical
development of its programs as well as the current capital raising market for biotechnology companies, the Company made the decision to
discontinue the Company sponsored trial for the invariant natural killer T-cell (“iNKT”) program and pause further patient
accrual to the Company sponsored adenosine trial for both PORT-6 and PORT-7. The Company is exploring strategic alternatives, which may
include finding a partner for one or more of its assets, a sale of the company, a merger, restructurings, both in and out of court, a
company wind down, further financing efforts or other strategic action.
Reverse Stock Split
The Company’s Board of Directors (the “Board”)
approved a reverse stock split of its ordinary shares at a ratio of 1-for-20. Beginning with the opening of trading on August 15, 2024,
the Company’s ordinary shares began trading on Nasdaq on a split-adjusted basis under the existing trading symbol “PRTG”.
The reverse stock split was implemented to increase the
per share trading price of the Company’s ordinary shares for the purpose of ensuring a share price high enough to comply with the
minimum $1.00 bid price requirement for continued listing on Nasdaq. The Company received notice from Nasdaq on August 30, 2024 informing
the Company that it had regained compliance with the minimum $1.00 bid price requirement for continued listing on Nasdaq.
As a result of the reverse stock split,
every twenty (20) pre-split ordinary shares were converted into one (1) post-split ordinary share. Any fractional shares resulting from
the reverse stock split were rounded up to the nearest whole post-split ordinary share. The reverse stock split affected all shareholders
uniformly and did not alter any shareholder’s percentage interest in the Company’s ordinary shares, except for adjustments
that may result from the treatment of fractional shares. All outstanding options and warrants entitling their holders to purchase the
Company’s ordinary shares were adjusted as a result of the reverse stock split, in accordance with the terms of each such security.
In addition, the number of ordinary shares reserved for future issuance pursuant to the Company’s equity incentive plans were also
appropriately adjusted. The number of authorized ordinary shares was not proportionately reduced because the Company has an unlimited
number of authorized ordinary shares available for issuance, as permitted under the laws of the British Virgin Islands.
All share and per share information
included in the condensed consolidated interim financial statements have been retroactively adjusted to reflect the impact of the reverse
stock split as if the stock split occurred at the beginning of the periods presented. The shares of ordinary shares authorized remained
at an unlimited number of ordinary shares without par value.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
As of September 30, 2024, the Company had cash and cash
equivalents of approximately $1.8
million and total current liabilities of approximately $0.9
million. For the six months ended September 30, 2024, the Company is reporting a net loss of approximately $3.0
million, and cash used in operating activities of approximately $3.3
million. As of November 25, 2024, the Company had approximately $1.7
million of cash and cash equivalents on hand.
The Company’s cash and cash equivalents balance
is decreasing, and the Company did not generate positive cash flows from operations for the six months ended September 30, 2024 and fiscal
year ended March 31, 2024 (“Fiscal 2024”).
In late Fiscal 2024, because of continued liquidity
constraints, the Company made the decision to discontinue further clinical development of its iNKT sponsored trial and pause further
patient accrual to its sponsored adenosine program for both PORT-6 and PORT-7. The Company is exploring strategic alternatives,
which may include finding a partner for one or more of its assets, a sale of our company, a merger, restructurings, both in and out
of court, company wind down, further financing efforts or other strategic action. These factors raise significant doubt about the
Company’s ability to continue as a going concern within one year after the date of the condensed consolidated interim
statement of financial position.
The Company has incurred significant operating losses
since inception and expects to continue to incur significant operating losses for the foreseeable future and may never become profitable.
The losses result primarily from its conduct of research and development activities. As previously discussed, the Company has discontinued
its iNKT sponsored trial and paused further patient accrual to the Company sponsored adenosine program in order to preserve cash resources.
Additionally, during the fourth quarter of Fiscal 2024, the Company sold its shares in Intensity on Nasdaq.
The Company historically has funded its operations principally
from proceeds from issuances of equity and debt securities. The Company will require significant additional capital to make the investments
it needs to execute its longer-term business plan, beyond the potential proceeds that could be reasonably generated from its “at-the-market”
(“ATM”) program and Committed Purchase Agreement (as defined below) with Lincoln Park Capital Fund, LLC (“Lincoln”)
given the Company’s current trading volume on Nasdaq. The Company’s ability to successfully raise sufficient funds through
the sale of debt or equity securities when needed is subject to many risks and uncertainties and, future equity issuances would result
in dilution to existing shareholders and any future debt securities may contain covenants that limit the Company's operations or ability
to enter into certain transactions. See Note 9, “Capital Stock and Reserves,” for a further discussion.
There can be no assurance that the Company’s evaluation
of strategic alternatives will result in any agreements or transactions, or that, if completed, any agreements or transactions will be
successful or on attractive terms. Any potential transaction would be dependent on a number of factors that may be beyond the Company’s
control, including, among other things, market conditions, industry trends, the interest of third parties in a potential transaction
with the Company and the availability of financing to the Company or third parties in a potential transaction with the Company on reasonable
terms. The process of reviewing strategic alternatives may require the Company to incur additional costs and expenses. It could negatively
impact the Company’s ability to attract, retain and motivate key employees, and expose the Company to potential litigation in connection
with this process or any resulting transaction. If the Company is unable to effectively manage the process, the Company’s financial
condition and results of operations could be adversely affected. In addition, any strategic alternative that may be pursued and completed
ultimately may not deliver the anticipated benefits or enhance shareholder value. There can be no guarantee that the process of evaluating
strategic alternatives will result in the Company entering into or completing a potential transaction within the anticipated timing or
at all. There is no set timetable for this evaluation and the Company does not intend to disclose developments with respect to this evaluation
unless and until the Company determines that further disclosure is appropriate or legally required. As of November 25, 2024, the Company
had approximately $1.7 million of cash and cash equivalents on hand, which the Company expects is only sufficient to cover its operating
needs through January 2025. These factors raise substantial doubt about the Company’s ability to continue as a going concern within
one year after the date of the condensed consolidated interim statement of financial position. There were no adjustments made to reflect
the effect of this doubt.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 3. BASIS OF PRESENTATION
Statement of Compliance and Basis of Presentation
These condensed consolidated interim financial statements
have been prepared in accordance with the International Financial Reporting Standards (“IFRS”) issued by the International
Accounting Standards Board (“IASB”), International Accounting Standards (“IAS”) 34 Interim Financial Reporting
and interpretations of the International Financial Reporting Interpretations Committee. These condensed consolidated interim financial
statements do not include all of the information required for full annual financial statements and should be read in conjunction with
the audited consolidated financial statements of the Company for the year ended March 31, 2024.
These condensed consolidated interim financial statements
have been prepared on an historical cost basis except for items disclosed herein at fair value (see Note 14, “Financial Instruments
and Risk Management”). In addition, these condensed consolidated interim financial statements have been prepared using the accrual
basis of accounting, except for cash flow information.
The Company has only one reportable operating segment.
These condensed consolidated interim financial statements
were approved and authorized for issuance by the Audit Committee (the “Audit Committee”) of the Board on November 26,
2024.
Consolidation
The consolidated financial statements include the accounts
of the Company and:
| (a) | SalvaRx, a wholly-owned subsidiary, incorporated on May 6, 2015 in the British Virgin Islands; |
| (b) | iOx, a wholly-owned subsidiary incorporated in the U.K. on February 10, 2015. In September 2021, the Company,
through SalvaRx, exchanged certain notes, accrued interest, warrants and receivables in exchange for shares of iOx representing 17.83%
of the outstanding shares of iOx. As a result of this exchange, the Company, through SalvaRx, increased its ownership of iOx from 60.49%
to 78.32%. On July 18, 2022, the Company purchased the remaining non-controlling interest of iOx. The Company’s 44% interest in
Stimunity S.A. (“Stimunity”) was transferred from Portage to iOx in December 2023 and was increased to 48.9% upon the conversion
of the convertible note to equity; |
| (c) | Saugatuck, a 70% owned subsidiary incorporated in the British Virgin Islands; |
| (d) | Tarus Therapeutic Holdings Ltd., a wholly-owned subsidiary incorporated in the British Virgin Islands; |
| (e) | PDS, a 100% owned subsidiary incorporated in Delaware, which provides human resources, and other services
to each operating subsidiary via a shared services agreement; |
| (f) | SalvaRx LLC, a wholly-owned subsidiary through SalvaRx incorporated in Delaware; |
| (g) | Saugatuck Rx LLC, a wholly-owned subsidiary of Saugatuck incorporated in Delaware; and |
| (h) | Tarus Therapeutics, LLC (“Tarus”), a wholly-owned subsidiary of Portage incorporated in Delaware. |
All inter-company balances and transactions have been
eliminated in consolidation.
Non-controlling interest in the equity of a subsidiary
is accounted for and reported as a component of shareholders’ equity. As of September 30, 2024 and March 31, 2024, non-controlling
interest represents the 30% shareholder ownership interest in Saugatuck and subsidiary, which is consolidated by the Company.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 3. BASIS OF PRESENTATION (Cont’d)
Functional and Presentation Currency
The Company’s functional and presentation currency
is the U.S. Dollar.
Use of Estimates and Judgments
The preparation of the condensed consolidated interim
financial statements in conformity with IFRS requires management to make judgments, estimates and assumptions that affect the application
of accounting policies and the reported amounts of assets, liabilities, income and expenses. Actual results may differ from these estimates.
Estimates and underlying assumptions are reviewed on
an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimates are revised and in any future
periods affected.
Significant areas where estimates are
made include valuation of financial instruments, deferred tax assets and liabilities, warrant liabilities, research and development costs,
contingent consideration assumed and measurement of share-based compensation. Significant areas where critical judgments are applied include
in-process research and development and warrant liabilities.
Reclassifications
Certain prior year amounts between
accounts payable and accrued liabilities have been reclassified for consistency with the current year presentation. These reclassifications
had no effect on the reported results of operations.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 4. SIGNIFICANT ACCOUNTING POLICIES
The accounting policies are set out in Note 4 to the
Company’s audited consolidated financial statements for Fiscal 2024. These policies have been applied consistently to all periods
presented in these condensed consolidated interim financial statements.
Recent Accounting Pronouncements
IFRS Pronouncements Issued
Impact of Adoption of Significant New IFRS Standards in Fiscal 2025
| (a) | Amendments to IAS 1: Non-current Liabilities with Covenants |
The amendments to IAS 1, “Presentation of Financial
Statements,” clarify how conditions with which an entity must comply within twelve months after the reporting period affect the
classification of a liability. The amendments also aim to improve information an entity provides related to liabilities subject to these
conditions.
The amendments to IFRS 16 and IAS 1 were effective for
annual periods beginning on or after January 1, 2024. The adoption of these amendments did not have a material effect on the Company’s
annual consolidated financial statements or the condensed consolidated interim financial statements for the three and six months ended
September 30, 2024.
| (b) | Amendments to IAS 7 and IFRS 7: Supplier Finance |
The amendments to IAS 7 and IFRS 7, “Statement
of Cash Flows,” require disclosures to enhance the transparency of supplier finance arrangements and their effects on an entity’s
liabilities, cash flows and exposure to liquidity risk. The amendments to IAS 7 and IFRS 7 were effective for annual periods beginning
on or after January 1, 2024 (with transitional reliefs in the first year). The adoption of these amendments did not have a material effect
on the Company’s annual consolidated financial statements or the condensed consolidated interim financial statements for the three
and six months ended September 30, 2024.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 4. SIGNIFICANT ACCOUNTING POLICIES (Cont’d)
New Accounting Standards, Interpretations and Amendments
Standards issued but not yet effective up to the date
of issuance of the Company’s condensed consolidated interim financial statements is listed below. This is of standards and interpretations
issued, which the Company reasonably expects to be applicable at a future date. The Company intends to adopt this standard when it becomes
effective.
Amendments to IAS 21 – Lack of Exchangeability
An entity is impacted by the amendments to IAS 21, “The
Effects of Changes in Foreign Exchange Rates,” when it has a transaction or an operation in a foreign currency that is not exchangeable
into another currency at a measurement date for a specified purpose. A currency is exchangeable when there is an ability to obtain the
other currency (with a normal administrative delay), and the transaction would take place through a market or exchange mechanism that
creates enforceable rights and obligations. The amendments to IAS 21 are effective for annual periods beginning on or after January 1,
2025, unless earlier adopted. The Company is evaluating whether the adoption of the amendments to IAS 21 would have a material effect
on the Company’s annual consolidated financial statements or its condensed consolidated interim financial statements.
NOTE 5. PREPAID EXPENSES AND OTHER RECEIVABLES
At September 30, 2024 and March 31, 2024, prepaid expenses
and other receivables were comprised of the following (in thousands):
| Schedule of prepaid expenses and other receivables | |
| | | |
| | |
| | |
September 30, 2024
(Unaudited) | |
March 31, 2024 |
| | |
| |
|
| Prepaid clinical research costs | |
$ | – | | |
$ | 1,924 | |
| Refund from Parexel | |
| 552 | | |
| – | |
| Prepaid insurance | |
| 249 | | |
| 575 | |
| Tax deposits | |
| 63 | | |
| 64 | |
| Other prepaid expenses | |
| 26 | | |
| 65 | |
| Other receivables | |
| 18 | | |
| 39 | |
| Security deposit on office lease expiring in May 2025 | |
| 14 | | |
| – | |
| Total prepaid expenses and other receivables | |
$ | 922 | | |
$ | 2,667 | |
The decrease in prepaid expenses and other receivables
from $2.7 million as of March 31, 2024 to $0.9 million as of September 30, 2024 is primarily
attributable to periodic amortization of prepaid clinical research costs and insurance premiums and the write-off of $1.1 million of advances
to Parexel International (IRL) Limited (“Parexel”), partially
offset by a $0.6 million refund receivable from Paraxel as of September 30, 2024. Refer to the “iOx – Parexel Master Services
Agreement” section of Note 12, “Commitments and Contingent Liabilities” for further discussion regarding this matter.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 6. WARRANT LIABILITY
On August 15, 2024, the Company effected
a reverse stock split of its ordinary shares at a ratio of 1-for-20. The number of warrants and exercise price information herein have
been retroactively adjusted to reflect the impact of the reverse stock split. See Note 1 “Nature of Operations” and Note 9,
“Capital Stock and Reserves” for further discussion on the reverse stock split.
The following table summarizes the changes in the warrant
liability during the three and six months ended September 30, 2024:
| Schedule of changes in the warrant
liability | |
| |
| |
|
| | |
Exercise Price | |
Warrants | |
Fair Value Balance |
| | |
| | | |
| | | |
| In 000’$ | |
| Warrant liability as of April 1, 2023 | |
$ | – | | |
| – | | |
$ | – | |
| Fair value of warrants at issuance on October 3, 2023: | |
| | | |
| | | |
| | |
| Class B Warrants | |
$ | 45.20 | | |
| 157,895 | | |
| 3,537 | |
| Class C Warrants | |
$ | 45.20 | | |
| 157,895 | | |
| 4,663 | |
| Placement Agent Warrants | |
$ | 47.50 | | |
| 7,896 | | |
| 232 | |
| Change in fair value of warrant liability | |
| | | |
| – | | |
| (6,868 | ) |
| Warrant liability as of March 31, 2024 | |
| – | | |
| 323,686 | | |
| 1,564 | |
| Change in fair value of warrant liability | |
| | | |
| – | | |
| (1,142 | ) |
| Warrant liability as of June 30, 2024 | |
| | | |
| 323,686 | | |
| 422 | |
| Change in fair value of warrant liability | |
| | | |
| – | | |
| 716 | |
| Warrant liability as of September 30, 2024 | |
| – | | |
| 323,686 | | |
$ | 1,138 | |
On September 29, 2023, the Company entered into a securities
purchase agreement (the “Purchase Agreement”) with an institutional and accredited investor in connection with a registered
direct offering (the “the Registered Direct Offering”) and a concurrent private placement (the “Private Placement”)
and together with the Registered Direct Offering, the “Offerings”). The Offerings closed on October 3, 2023.
Pursuant to the Purchase Agreement, in the Registered
Direct Offering, the Company sold (i) 98,500 shares of the Company’s ordinary shares at a purchase price of $38.00 per share and
(ii) pre-funded warrants (the “Pre-Funded Warrants”) to purchase up to 59,395 ordinary shares, at a purchase price of $37.98
per Pre-Funded Warrant. All Pre-Funded Warrants, which were exercisable for one ordinary share at an exercise price of $0.02 per share,
were exercised in full on May 29, 2024.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 6. WARRANT LIABILITY (Cont’d)
In the Private Placement, the Company issued to such
institutional and accredited investor unregistered warrants to purchase up to 157,895 ordinary shares (the “Series A Warrants”),
unregistered warrants to purchase up to 157,895 ordinary shares (the “Series B Warrants”), and unregistered warrants to purchase
up to 157,895 ordinary shares (the “Series C Warrants,” together with the Series A Warrants and the Series B Warrants, the
“Private Warrants”), together exercisable for an aggregate of up to 473,685 ordinary shares (the “Private Warrant Shares”).
Pursuant to the terms of the Purchase Agreement, for each ordinary share and Pre-Funded Warrant issued in the Registered Direct Offering,
an accompanying Series A Warrant, Series B Warrant and Series C Warrant were issued to such institutional and accredited investor. Each
Series A Warrant is exercisable for one Private Warrant Share at an exercise price of $38.00 per share, is immediately exercisable and
will expire 18 months from the date of issuance. Each Series B Warrant is exercisable for one Private Warrant Share at an exercise price
of $45.20 per share, is immediately exercisable and will expire three years from the date of issuance. Each Series C Warrant is exercisable
for one Private Warrant Share at an exercise price of $45.20 per share, is immediately exercisable and will expire five years from the
date of issuance. The net proceeds to the Company from the Offerings were approximately $5.3 million, after deducting placement agent’s
fees and estimated offering expenses of approximately $0.7 million.
Pursuant to an engagement letter, dated as of August
26, 2023, between the Company and H.C. Wainwright & Co., LLC (the “Placement Agent”), the Company paid the Placement Agent
a total cash fee equal to 6.0% of the aggregate gross proceeds received in the Offerings, or $0.36 million. The Company also paid the
Placement Agent in connection with the Offerings a management fee equal to 1.0% of the aggregate gross proceeds raised in the Offerings
($0.06 million), $75,000 for non-accountable expenses and $15,950 for clearing fees. In addition, the Company issued to the Placement
Agent, or its designees, warrants to purchase up to 7,896 ordinary shares (the “Placement Agent Warrants,” and together with
the Pre-Funded Warrants and the Private Warrants, the “Warrants”), which represented 5.0% of the aggregate number of ordinary
shares and Pre-Funded Warrants sold in the Registered Direct Offering. The Placement Agent Warrants have substantially the same terms
as the Series B Warrants and the Series C Warrants, except that the Placement Agent Warrants have an exercise price equal to $47.50, or
125% of the offering price per ordinary share sold in the Registered Direct Offering and will be exercisable for five years from the commencement
of the sales pursuant to the Offerings. The Private Warrants, Private Warrant Shares, Placement Agent Warrants and ordinary shares underlying
the Placement Agent Warrants were registered for resale under the Securities Act of 1933, as amended (the “Securities Act”)
pursuant to a registration statement on Form F-1 that was declared effective by the SEC on November 7, 2023 (the “Resale Registration
Statement”).
The Series B Warrants, the Series C Warrants and the
Placement Agent Warrants include the obligation, in the event of a Fundamental Transaction, as defined in the Series B Warrants, the Series
C Warrants and the Placement Agent Warrants, for the Company or the successor entity to purchase the warrants from the holder at the discretion
of the holder and at the Black-Scholes value, as defined in the warrant agreements. As a result, management concluded that, in line with
IAS 9, “Financial Instruments” and IAS 32, “Financial Instruments: Presentation,” such warrants will be accounted
for as financial liabilities on the condensed consolidated interim statement of financial position with the changes in fair value recognized
in the condensed consolidated interim statement of operations and other comprehensive income (loss). The Company allocated the net proceeds
of $5.3 million for the Registered Direct Offering to the warrant liability and recognized the excess of the fair value of the warrant
liabilities at inception of $3.1 million as a loss on the Registered Direct Offering of $2.4 million and offering expenses of $0.7 million.
The Company also recorded a loss of $0.7 million and a gain of $0.4 million from the change in the fair value of the warrant liabilities
for the three and six months ended September 30, 2024, respectively, resulting in a warrant liability balance of $1.1 million at September
30, 2024.
The Company filed the Resale Registration Statement to
register for the resale of the Private Warrant Shares and the ordinary shares issuable upon the exercise of the Placement Agent Warrants,
which was declared effective by the SEC on November 7, 2023. Pursuant to the terms of the Purchase Agreement, the Company is obligated
to use its commercially reasonable efforts to keep the Resale Registration Statement effective at all times until such institutional and
accredited investor (and its successors and assigns) no longer owns any Private Warrants or ordinary shares issuable upon exercise thereof.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 6. WARRANT LIABILITY (Cont’d)
On May 29, 2024, Armistice Capital Master Fund Ltd. exercised
its Pre-Funded Warrants at an exercise price of $0.02 per share increasing the Company’s total shares outstanding to 1,048,765.
The accounting for the Series A Warrants and the Pre-Funded
Warrants is detailed below in Note 9, “Capital Stock and Reserves.”
Series B Warrants
A fair value of
$22.40 per each Series B Warrant was identified at the issue date of October 3, 2023. A fair value of $3.05 per each warrant has been
identified as of September 30, 2024.
The inputs associated
with calculating the fair value are reflected below.
| Schedule of fair value | |
| |
|
| | |
October 3, 2023 | |
September 30, 2024 |
| Exercise price | |
$45.20 | |
$45.20 |
| Share price | |
$39.40 | |
$6.65 |
| Expected life | |
3.01 years | |
2.01 years |
| Expected volatility | |
90.4% | |
159.22% |
| Risk-free interest rate | |
4.95% | |
3.66% |
| Dividend yield | |
– | |
– |
Series C Warrants
A fair value of
$29.60 per each Series C Warrant was identified at the issue date of October 3, 2023. A fair value of $3.91 per each warrant has been
identified as of September 30, 2024.
The inputs associated
with calculating the fair value are reflected below.
| Schedule of fair value | |
| |
|
| | |
October 3, 2023 | |
September 30, 2024 |
| Exercise price | |
$45.20 | |
$45.20 |
| Share price | |
$39.40 | |
$6.65 |
| Expected life | |
5.00 years | |
4.01 years |
| Expected volatility | |
100.7% | |
129.26% |
| Risk-free interest rate | |
4.80% | |
3.58% |
| Dividend yield | |
– | |
– |
Placement Agent Warrants
A fair value of
$29.40 per each Placement Agent Warrant was identified at the issue date of October 3, 2023. A fair value of $3.86 per each warrant has
been identified as of September 30, 2024.
The inputs associated
with calculating the fair value are reflected below.
| Schedule of fair value | |
| |
|
| | |
October 3, 2023 | |
September 30, 2024 |
| Exercise price | |
$47.60 | |
$47.50 |
| Share price | |
$39.40 | |
$6.65 |
| Expected life | |
4.99 years | |
4.01 years |
| Expected volatility | |
100.7% | |
129.26% |
| Risk-free interest rate | |
4.80% | |
3.58% |
| Dividend yield | |
– | |
– |
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 7. ACCOUNTS PAYABLE AND ACCRUED LIABILITIES
At September 30, 2024 and March 31, 2024, accounts payable
and accrued liabilities were comprised of the following (in thousands):
| Schedule of accounts payable and accrued liabilities | |
| | | |
| | |
| | |
September 30, 2024
(Unaudited) | |
March 31, 2024 |
| Accounts payable and amounts accrued to CRO | |
$ | 49 | | |
$ | 1,413 | |
| Accrued bonuses and other payroll-related expenses | |
| 371 | | |
| 525 | |
| Accrued accounting and auditing fees | |
| 127 | | |
| 189 | |
| Accrued legal fees | |
| 54 | | |
| 194 | |
| Accrued other professional fees | |
| 33 | | |
| 157 | |
| Other accounts payable | |
| 172 | | |
| 136 | |
| Accrued clinical and R&D services | |
| 3 | | |
| 179 | |
| Other | |
| 36 | | |
| 43 | |
| Total accounts payable and accrued liabilities | |
$ | 845 | | |
$ | 2,836 | |
The decrease in accounts
payable and accrued liabilities from $2.8 million as of March 31, 2024 to $0.8 million as of September 30, 2024 is primarily attributable
to the write-off of $1.486 million in connection with the settlement with Parexel. Refer to the “iOx – Parexel Master
Services Agreement” section of Note 12, “Commitments and Contingent Liabilities” for further discussion regarding this
matter.
NOTE 8. INCOME TAXES
The Company is a BVI business company. The BVI government
does not, under existing legislation, impose any income or corporate tax on corporations.
PDS is a U.S. corporation and is subject to U.S. federal,
state and local income taxes, as applicable.
iOx is subject to U.K. taxes.
The following is a reconciliation of the U.S. taxes to
the effective income tax rates for the six months ended September 30, 2024 and 2023 (in thousands, except percentages):
| Schedule of effective income tax rates | |
| | | |
| | |
| | |
Six Months Ended September 30, |
| | |
2024 | |
2023 |
| Loss before income taxes | |
$ | 3,024 | | |
$ | 12,143 | |
| Income tax benefit | |
$ | 3 | | |
$ | 1,052 | |
| Effective tax rate | |
| 0.10 | % | |
| 8.66 | % |
The Company’s effective tax rates were 0.10% and
8.66% for the six months ended September 30, 2024 and 2023, respectively. The decrease in the effective tax rate was primarily due to
a change in the mix of income and losses in the various tax jurisdiction in which the Company operates.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 9. CAPITAL STOCK AND RESERVES
Authorized ordinary shares: Unlimited number of
Portage ordinary shares without par value.
On August 15, 2024, the Company effected a reverse
stock split of its ordinary shares at a ratio of 1-for-20. The reverse stock split decreased the number of ordinary shares
outstanding as of September 30, 2023, from 17,808,225
to 890,412.
All share and per share information included in the condensed consolidated interim financial statements and in the Notes to the
condensed consolidated interim financial statements have been retroactively adjusted to reflect the impact of the reverse stock
split unless otherwise noted. The number of ordinary shares authorized remained at an unlimited number of common shares without par value.
Any fractional shares resulting from the reverse
stock split were rounded up to the nearest whole post-split ordinary share. As a result of this round up provision, the Company
issued an additional 52,254 shares.
Portage filed a shelf registration
statement with the SEC in order to sell ordinary shares, debt securities, warrants and units in one or more offerings from time to time,
which became effective on March 8, 2021 (“March 2021 Registration Statement”). In connection with the March 2021 Registration
Statement, Portage has filed with the SEC:
| · | a base prospectus, which covered the offering, issuance and sale by Portage of up
to $200 million in the aggregate of the securities identified above from time to time in one or more offerings; |
| · | a prospectus supplement, which covered the offer, issuance and sale by Portage in
its ATM offering of up to a maximum aggregate offering price of $50 million of Portage’s ordinary shares that may be issued and
sold from time to time under a Controlled Equity Offering Sales Agreement, dated February 24, 2021 (the “Sales Agreement”),
with Cantor Fitzgerald & Co., the sales agent (“Cantor Fitzgerald”); |
| · | a prospectus supplement dated June 24, 2021, for the offer, issuance and sale by
Portage of 57,500 ordinary shares for gross proceeds of approximately $26.5 million in a firm commitment underwritten public offering
with Cantor Fitzgerald; |
| · | a prospectus supplement dated August 19, 2022, for the resale of up to $30 million
in ordinary shares that Portage may sell from time to time to Lincoln and an additional 4,726 shares that were issued to Lincoln; and |
| · | a prospectus supplement dated September 29, 2023 for the offer, issuance and
sale by Portage in a registered direct public offering through H.C. Wainwright & Co., the placement agent, to an institutional
and accredited investor of (i) 98,500 ordinary shares at a purchase price of $38.00 per share; and (ii) Pre-Funded Warrants to
purchase up to 59,395 ordinary shares, at a purchase price of $37.98 per Pre-Funded Warrant Shares, for aggregate gross proceeds of
approximately $6 million. All Pre-Funded Warrants, which were exercisable
for one ordinary share at an exercise price of $0.02 per share, were exercised in full on May 29, 2024. |
The Sales Agreement permits the Company
to sell in an ATM program up to $50 million of ordinary shares from time to time. Through September 30, 2024, the Company raised approximately $4.2 million in gross proceeds through the sale of shares of common shares
under the ATM program. Any sales under the prospectus will be deemed to be
made pursuant to an ATM program as defined in Rule 415(a)(4) promulgated under the Securities Act.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 9. CAPITAL STOCK AND RESERVES (Cont’d)
On July 6, 2022, the Company entered into a Purchase
Agreement (the “Committed Purchase Agreement”) with Lincoln, under which it may require Lincoln to purchase ordinary shares
of the Company having an aggregate value of up to $30 million (the “Purchase Shares”) over a period of 36 months. Upon execution
of the Committed Purchase Agreement, the Company issued to Lincoln 4,726 ordinary shares, representing a 3% commitment fee. Pursuant to
the Committed Purchase Agreement, Lincoln will be obligated to purchase the Purchase Shares in three different scenarios that are based
on various market criteria and share amounts. The Company has the right to terminate the Committed Purchase Agreement for any reason,
effective upon one business day prior written notice to Lincoln. Lincoln has no right to terminate the Committed Purchase Agreement. The
requirement that Lincoln must make a purchase will be suspended based on various criteria such as there not being an effective registration
statement for Lincoln to be able to resell the ordinary shares it is committed to purchase and market criteria such as the Company continuing
to be Depository Trust Company eligible, among other things. The Committed Purchase Agreement does not impose any financial or business
covenants on the Company, and there are no limitations on the use of proceeds. The Company may raise capital from other sources in its
sole discretion; provided, however, that the Company shall not enter into any similar agreement for the issuance of variable priced equity-like
securities until the three-year anniversary of the date of the Committed Purchase Agreement, excluding, however, an ATM transaction with
a registered broker-dealer, which includes any sales under the Sales Agreement with Cantor Fitzgerald.
During the six months ended September 30, 2023, the
Company sold 9,331
ordinary shares under the ATM program, generating net proceeds of approximately $0.7
million. There were no shares sold under the ATM program during the six months ended September 30, 2024.
The Company’s March 2021 Registration Statement
expired on March 8, 2024. In order to issue additional shares under its ATM program or the Committed Purchase Agreement in the future,
the Company would be required to file a new registration statement, which must be declared effective by the SEC prior to use, and to file
a prospectus supplement related to the ATM program and the Committed Purchase Agreement, as the case may be.
Furthermore, the ATM program and the Committed Purchase
Agreement with Lincoln are generally limited based on, among other things, the Company’s Nasdaq trading volume. Under General Instruction
I.B.5 to Form F-3 (the “Baby Shelf Rule”), the amount of funds the Company can raise through primary public offerings of securities
in any 12-month period using a registration statement on Form F-3 is limited to one-third of the aggregate market value of the ordinary
shares held by the Company’s non-affiliates, which limitation may change over time based on its stock price, number of ordinary
shares outstanding and the percentage of ordinary shares held by non-affiliates. Accordingly, the Company is limited by the Baby Shelf
Rule as of the filing of this Form 6-K, until such time as its non-affiliate public float exceeds $75 million.
On September 29, 2023, the Company entered into the Purchase
Agreement with an institutional and accredited investor in connection with the Registered Direct Offering and the Private Placement. The
Offerings closed on October 3, 2023.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 9. CAPITAL STOCK AND RESERVES (Cont’d)
The Company filed the Resale Registration Statement to
register for resale the Private Warrant Shares and the ordinary shares issuable upon the exercise of Placement Agent Warrants, which was
declared effective by the SEC on November 7, 2023. Pursuant to the terms of the Purchase Agreement, the Company is obligated to use its
commercially reasonable efforts to keep the Resale Registration Statement effective at all times until such institutional and accredited
investor (and its successors and assigns) no longer owns any Private Warrants or ordinary shares issuable upon exercise thereof.
If a Fundamental Transaction (as defined in the Warrants)
occurs, then the successor entity will succeed to, and be substituted for the Company, and may exercise every right and power that the
Company may exercise and will assume all of the Company’s obligations under the Warrants with the same effect as if such successor
entity had been named in the Warrants themselves. If holders of ordinary shares are given a choice as to the securities, cash or property
to be received in such a Fundamental Transaction, then the holders of the Warrants shall be given the same choice as to the consideration
they would receive upon any exercise of the Warrants following such a Fundamental Transaction. Additionally, as more fully described in
the Series B Warrants, Series C Warrants and Placement Agent Warrants, in the event of certain Fundamental Transactions, the holders of
the Series B Warrants, Series C Warrants and Placement Agent Warrants will be entitled to receive cash consideration in an amount equal
to the Black-Scholes value of the Series B Warrants, Series C Warrants and Placement Agent Warrants, as the case may be, upon the consummation
of such Fundamental Transaction.
See Note 6, “Warrant Liability,” for a discussion
of the Registered Direct Offering.
Series A Warrants and Pre-Funded Warrants
The Series A Warrants and the Pre-Funded Warrants are
classified as a component of equity because they are freestanding financial instruments that are legally detachable and separately exercisable
from the ordinary shares with which they were issued, are immediately exercisable, do not embody an obligation for the Company to repurchase
its shares, and permit the holders to receive a fixed number of ordinary shares upon exercise. In addition, the Series A Warrants and
the Pre-Funded Warrants do not provide any guarantee of value or return.
On the October 3, 2023
issue date, the calculated fair value of the Series A Warrants and the Pre-Funded Warrants as of March 31, 2024 was $2.968 million ($0.94
per such warrant). Because the fair value of the warrants accounted for as liabilities exceeded the net proceeds from the Registered
Direct Offering, the proceeds allocated to the Common Shares, the Pre-Funded Warrants and the Series A warrants was zero.
Series A Warrants
The inputs associated
with calculating the fair value are reflected below.
| Schedule of fair value | |
| |
| | |
October 3, 2023 |
| Exercise price | |
$38.00 | |
| Share price | |
$39.40 | |
| Expected life | |
1.50 years | |
| Expected volatility | |
96.0% | |
| Risk-free interest rate | |
5.32% | |
| Dividend yield | |
– | |
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 9. CAPITAL STOCK AND RESERVES (Cont’d)
Nature and Purpose of Reserves
Stock Option Reserve
The stock option reserve reflects the reserve of compensation
expense recognized over the vesting period based upon the grant date fair value of the Company’s equity settled grants calculated
in accordance with IFRS 2, “Share-based Payment”. See Note 10, “Stock Option Reserve,” below for a further discussion.
NOTE 10. STOCK OPTION RESERVE
Amended and Restated 2021 Equity Incentive
Plan and Grants of Stock Options and Restricted Stock Units
The following table provides the activity for the Company’s
stock option reserve for the six months ended September 30, 2024 and 2023 (in thousands):
| Schedule of stock options | |
| | | |
| | |
| | |
Six Months Ended September 30, |
| | |
2024 | |
2023 |
| Balance, beginning of period | |
$ | 23,841 | | |
$ | 21,204 | |
| Share-based compensation expense | |
| 285 | | |
| 1,512 | |
| Balance, end of period | |
$ | 24,126 | | |
$ | 22,716 | |
| | |
| | | |
| | |
On August 15, 2024, the Company effected
a reverse stock split of its ordinary shares at a ratio of 1-for-20. All share and per share information have been retroactively adjusted
to reflect the impact of the reverse stock split unless otherwise noted. See Note 1 “Nature of Operations” and Note 9, “Capital
Stock and Reserves” for further discussion on the reverse stock split.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 10. STOCK OPTION RESERVE (Cont’d)
As of September 30, 2024, 82,429 shares were reserved
for awards previously granted and 59,997 shares were available for future awards under the Amended and Restated 2021 Equity Incentive
Plan.
The changes in the number of stock options issued and
outstanding for the six months ended September 30, 2024 and 2023 were as follows:
| Schedule of changes in the number of stock options | |
| | | |
| | |
| | |
Six Months Ended September 30, |
| | |
2024 | |
2023 |
| Balance, beginning of period | |
| 90,281 | | |
| 98,171 | |
| Expired or forfeited | |
| (25,167 | ) | |
| – | |
| Balance, end of period | |
| 65,114 | | |
| 98,171 | |
| Exercisable, end of period | |
| 43,723 | | |
| 38,461 | |
The table above does not include fully vested
restricted stock units (“RSUs”) granted to certain executives in January 2021 and January 2022. In January 2024, the
company issued 308
shares for the exercise of 470
RSUs net of 162
shares repurchased into treasury to pay for an employee’s payroll taxes. In September 2024, the company issued 882
shares for the exercise of 1,152
RSUs net of 270
shares repurchased into treasury to pay for an employee’s payroll taxes. As September 30, 2024, there were 17,315
RSUs granted and outstanding of which 12,150 RSUs
expire in January 2031 and 5,165 expire in January 2032.
The following is the weighted average exercise price
and the remaining contractual life for outstanding options as of September 30,
2024 and 2023:
| Schedule of weighted average exercise price
and the remaining contractual life | |
| | | |
| | |
| | |
As of September 30, |
| | |
2024 | |
2023 |
| | |
PBI Amended and Restated
2021 Equity Incentive Plan |
| Weighted average exercise price | |
$ | 205.18 | | |
| 210.60 | |
| Weighted average remaining contractual life (in years) | |
| 7.39 | | |
| 8.36 | |
The vested options can be exercised at any time in accordance
with the applicable option agreement. The exercise price was greater than the market price for all options outstanding as of September
30, 2024 and March 31, 2024.
The Company recorded approximately $0.141 million and
$0.743 million of share-based compensation expense with respect to the Amended and Restated 2021 Equity Incentive Plan in the three months
ended September 30, 2024 and 2023, respectively. The Company recorded approximately $0.285 million and $1.512 million of share-based compensation
expense with respect to the Amended and Restated 2021 Equity Incentive Plan in the six months ended September 30, 2024 and 2023, respectively.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 11. LOSS PER SHARE
Basic earnings per
share (“EPS”) is calculated by dividing the net loss attributable to ordinary equity holders of the Company by the weighted
average number of ordinary shares outstanding during the period.
Diluted EPS is calculated
by dividing the net loss attributable to ordinary equity holders of the Company by the weighted average number of ordinary shares outstanding
during the period plus the weighted average number of ordinary shares that would be issued on conversion of all the dilutive potential
ordinary shares into ordinary shares. Shares issuable under Pre-Funded Warrants are considered outstanding for this purpose.
The calculation
of Basic and Diluted EPS reflects the Pre-Funded Warrants as outstanding shares.
The following table reflects the loss and share data
used in the basic and diluted EPS calculations (in thousands, except per share amounts):
| Schedule of basic and diluted EPS calculations | |
| | | |
| | | |
| | | |
| | |
| | |
Three Months Ended September 30, | |
Six Months Ended September 30, |
| | |
2024 | |
2023 | |
2024 | |
2023 |
| Numerator | |
| |
| |
| |
|
| Net loss attributable to owners of the Company | |
$ | (1,360 | ) | |
$ | (5,158 | ) | |
$ | (3,016 | ) | |
$ | (11,077 | ) |
| Denominator | |
| | | |
| | | |
| | | |
| | |
| Weighted average number of shares – Basic and Diluted | |
| 1,076 | | |
| 890 | | |
| 1,063 | | |
| 888 | |
| Basic and diluted loss per share | |
$ | (1.26 | ) | |
$ | (5.80 | ) | |
$ | (2.84 | ) | |
$ | (12.47 | ) |
Number of ordinary shares are retroactively adjusted
to reflect the impact of the 1-for-20 reverse stock split on August 15, 2024.
The inclusion of the Company’s share purchase warrants
(other than Pre-Funded Warrants, as described above), stock options and RSUs in the computation of diluted loss per share would have an
anti-dilutive effect on loss per share and are therefore excluded from the computation. Consequently, there is no difference between basic
loss per share and diluted loss per share for the three and six months ended September 30, 2024 and 2023. The following table reflects
the Company’s outstanding securities by year that would have an anti-dilutive effect on loss per share and, accordingly, were excluded
from the calculation.
| Schedule of anti-dilutive effect on loss per share | |
| | | |
| | |
| | |
As of September 30, |
| | |
2024 | |
2023 |
| Warrants | |
| 481,581 | | |
| – | |
| Stock options | |
| 65,114 | | |
| 98,171 | |
| Restricted stock units | |
| 17,315 | | |
| 18,937 | |
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 12. COMMITMENTS AND CONTINGENT LIABILITIES
iOx – Parexel Master Services Agreement
Effective March 15, 2022, iOx entered into a Master Services
Agreement (the “MSA”) with Parexel under which Parexel agreed to act as
a clinical service provider (“CRO”) pursuant to a work order (“Work Order”) effective June 1, 2022. Under such Work Order,
Parexel planned to operate a Phase 2 trial of IMM60 and pembrolizumab in advanced melanoma and non-small lung cancer. The MSA provided
for a five-year term, and the Work Order provided for a term to end upon the completion of the services required. The budget provided
for service fees and pass-through expenses and clinical sites totaling $11.5 million. During the year ended March 31, 2023 (“Fiscal
2023”), the Company executed two change orders resulting in a $0.6 million increase in the overall estimated budgeted costs. As
a result of the Company’s decision to discontinue the development with respect to this program, on December 20, 2023, the Company
provided Parexel notice of termination of the contract, with a planned termination date of April 18, 2024.
As the ongoing CRO services were wound down, the Company
noticed that expenses incurred under the CRO agreement were higher than originally budgeted. Parexel agreed to refund the Company $0.552
million and release the liability for amounts invoiced and unbilled services totaling $1.486 million, and the Company forfeited advanced
payments of $1.091 million. As a result, the Company recognized a net gain of $0.946 million as of September 30, 2024. As of March 31,
2024, the Company reflected $1.414 million of payables to Parexel in accounts payable and accrued liabilities and $1.277 million in prepaid
expenses and other receivables in the consolidated statement of financial position. The Company
received the refund from Parexel on October 3, 2024.
Tarus – Fortrea Clinical Service Agreement
On March 1, 2023, Tarus entered into a clinical service
agreement with Fortrea Inc. (formerly Labcorp Drug Development Inc.), a third-party CRO. The term of the agreement is through the earlier
of August 14, 2025 or the completion of provision of services and the payment of contractual obligations. The budgeted costs for the services
to be provided is approximately $12.1 million. Because of the Company’s decision to discontinue and pause further accrual of all
clinical studies, the Company is negotiating a revision to the services required under the change in circumstances.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 13. RELATED PARTY TRANSACTIONS
SalvaRx Acquisition
Two of the Company’s directors are also directors
of SalvaRx Group plc, a company which owns approximately 4.1% of the Company’s issued and outstanding ordinary shares as of September
30, 2024.
Investments
The Company has entered into related party transactions
and certain services agreements with its investees. Key management personnel of the Company have also entered into related party
transactions with investees. Key management personnel are those people who have the authority and responsibility for planning, directing
and controlling the activities of the Company, including directors and senior management of the Company.
The following subsidiaries and associates are considered
related parties:
| (a) | Stimunity. The Chief Executive Officer (“CEO”) of Portage is one of three members of
the board of directors of Stimunity. The Company wrote-off its investment in Stimunity to nil in the Fiscal 2024 period. |
| (b) | iOx. Upon execution of the iOx Share Exchange on July 18, 2022, the non-Portage director resigned
from the iOx board leaving two Portage insiders as directors. The CEO of Portage is also the CEO of iOx, and the management team of Portage
comprises the management team of iOx. |
| (c) | Saugatuck. The Chairman and CEO of the Company is the sole director of Saugatuck. Saugatuck is
70% owned by the Company and is controlled by Portage. |
| (d) | Intensity. The CEO of Portage previously served as a part-time officer of Intensity until becoming
a consultant in 2023. Additionally, Intensity provided services (primarily rent) to Portage through April 2023. For the six months ended
September 30, 2023, the Company paid $0.01 million. No such services were provided subsequent to April 2023. |
| (e) | Portage Development Services Inc. PDS provides human resources and other services to each operating
subsidiary of Portage through shared services agreements. |
Transactions between the parent company and its subsidiaries,
which are related parties, have been eliminated in consolidation and are not disclosed in this note.
Board Resignations and Appointments
On April 25, 2024, Mark Simon resigned
all of his positions on the Board of Directors, and on April 26, 2024, Linda Kozick and Dr. Robert Glassman resigned all of their positions
on the Board of Directors. On April 30, 2024, Dr. Jean -Christophe Renondin and Dr. Justin Stebbing were elected to the Board.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 13. RELATED PARTY TRANSACTIONS (Cont’d)
Bonuses & Board Compensation
Arrangements
In December 2022,
the Board approved executive performance bonuses, as recommended by the Compensation Committee, totaling $0.6 million, which is equivalent
to 73.5% of original annual targets established by the Board in December 2021. The bonuses were approved based upon the original performance
targets established. The Board further approved a payment structure of 25% of approved bonuses, which were paid in January 2023, with
the balance of amounts due payable upon a new financing. The accrued, unpaid amount of approximately $0.4 million is included in accounts
payable and accrued liabilities in the condensed consolidated interim statements of financial position as of each of September 30, 2024
and March 31, 2024. No executive performance bonus has been approved by the Compensation Committee or the Board for Fiscal Year
2024.
Effective January 1, 2022, each non-employee Board
member is entitled to receive cash Board fees of $40,000 per annum, payable quarterly in arrears. Additionally, each non-employee
Board member is entitled to an annual grant of 345 options to purchase Portage ordinary shares, which would vest the first annual
anniversary of the grant date. The Company incurred Board fees totaling nil 0 and $82,500 during the three months ended September
30, 2024 and 2023, respectively, and $7,205 and $165,000 during the six months ended September 30, 2024 and 2023, respectively.
Non-employee Board chairpersons are entitled to an annual
cash fee of $30,000, payable quarterly in arrears. In lieu of a non-executive chairperson, the lead director is entitled to an annual
cash fee of $20,000 per annum paid quarterly in arrears. Additionally, the chairperson of each of the Audit Committee, Compensation Committee
and Nominating Committee of the Board is entitled to annual fees of $15,000, $12,000 and $8,000, respectively, payable quarterly in arrears.
Members of those committees are entitled to annual fees of $7,500, $6,000 and $4,000, respectively, payable quarterly in arrears. All
non-executive board members waived their right to board fees for the six months ended September 30, 2024, except for the non-executive
board members who resigned in late April 2024.
Retention Agreements and General Releases
On July 22, 2024, the Company and Portage
Development Services, Inc. entered into a Retention Agreement and General Release (“Retention Agreement”) with each of Allan
Shaw (“Employee”) and Joseph Ciavarella (“Consultant”). Under the terms of each of the Retention Agreements, Employee’s
current employment agreement and Consultant’s current consulting agreement both terminated on July 22, 2024, except with respect
to certain provisions. In return for continuing to provide services to the Company and its affiliates and completing certain Applicable
Retention Events, as defined in the respective Retention Agreements, the Company paid an aggregate $0.2 million (the “Retention
Amount”) to Employee and Consultant, in addition to their monthly pay through September 30, 2024.
The Company accrued the Retention
Amount, as well as the monthly pay totaling $0.2 million through September 30, 2024, in the condensed consolidated interim financial statements
for the three months ended June 30, 2024, as the material terms of the Retention Agreements were known and agreed upon at June 30, 2024.
In accordance with the terms of the Retention Agreements, the Company paid in full the Retention Amount and issued 14,348 ordinary shares
to the Employee and Consultant in lieu of cash bonuses accrued in Fiscal 2023 totaling $0.1 million by September 30, 2024, the date on
which the Employee’s employment and the Consultant’s consulting relationship with the Company and its affiliates ended.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 14. FINANCIAL INSTRUMENTS AND RISK MANAGEMENT
The Company’s financial instruments recognized
in the Company’s condensed consolidated interim statements of financial position consist of the following:
Fair value estimates are made at a specific point in
time, based on relevant market information and information about financial instruments. These estimates are subject to and involve uncertainties
and matters of significant judgment; and therefore, these estimates cannot be determined with precision. Changes in assumptions could
significantly affect these estimates.
The following table summarizes the Company’s financial
instruments as of September 30, 2024 and March 31, 2024 (in thousands):
| Schedule of financial instruments | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
September 30, 2024
(Unaudited) | |
March 31, 2024 |
| | |
Amortized
Cost | |
FVTOCI | |
FVTPL | |
Amortized
Cost | |
FVTOCI | |
FVTPL |
| Financial assets | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Cash and cash equivalents | |
$ | 1,764 | | |
$ | – | | |
$ | – | | |
$ | 5,028 | | |
$ | – | | |
$ | – | |
| Prepaid expenses and other current assets | |
$ | 922 | | |
$ | – | | |
$ | – | | |
$ | 2,667 | | |
$ | – | | |
$ | – | |
| | |
| | | |
| | | |
| | | |
| | |
| | |
September 30, 2024
(Unaudited) | |
March 31, 2024 |
| | |
Amortized
Cost | |
FVTPL | |
Amortized
Cost | |
FVTPL |
| Financial liabilities | |
| | | |
| | | |
| | | |
| | |
| Accounts payable and accrued liabilities | |
$ | 845 | | |
$ | – | | |
$ | 2,836 | | |
$ | – | |
| Warrant liability | |
$ | – | | |
$ | 1,138 | | |
$ | – | | |
$ | 1,564 | |
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 14. FINANCIAL INSTRUMENTS AND RISK MANAGEMENT (Cont’d)
A summary of the Company’s risk exposures as it
relates to financial instruments are reflected below.
Fair value of Financial Instruments
The Company’s financial assets and liabilities
are comprised of cash and cash equivalents, receivables and investments in equities and public entities, accounts payable and accrued
liabilities, lease liability, and warrant liability.
The Company classifies the fair value of these transactions
according to the following fair value hierarchy based on the amount of observable inputs used to value the instrument:
| · | Level 1 – Values are based on unadjusted quoted prices available in active markets for identical
assets or liabilities as of the reporting date. |
| · | Level 2 – Values are based on inputs, including quoted forward prices for commodities, time value
and volatility factors, which can be substantially observed or corroborated in the marketplace. Prices in Level 2 are either directly
or indirectly observable as of the reporting date. |
| · | Level 3 – Values are based on prices or valuation techniques that are not based on observable market
data. Investments are classified as Level 3 financial instrument. |
Assessment of the significance of a particular input
to the fair value measurement requires judgment and may affect the placement within the fair value hierarchy.
Management has assessed that the fair values of cash
and cash equivalents, other receivables and accounts payable approximate their carrying amounts largely due to the short-term maturities
of these instruments.
The following methods and assumptions were used to estimate
their fair values:
Warrant Liability: The fair value is estimated
using a Black-Scholes model and in certain cases, a Monte Carlo simulation (Level 3) (see Note 6, “Warrant Liability”).
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 14. FINANCIAL INSTRUMENTS AND RISK MANAGEMENT (Cont'd)
Credit Risk, Liquidity Risk and Foreign Currency Risk
Our financial instruments are exposed to certain financial risks: Credit Risk, Liquidity
Risk and Foreign Currency Risk.
Credit Risk
Credit risk is the risk of loss associated with a counterparty’s
inability to fulfill its payment obligations. The credit risk is attributable to various financial instruments, as noted below. The credit
risk is limited to the carrying value as reflected in the Company’s condensed consolidated interim statements of financial position.
Cash and cash equivalents: Cash and cash equivalents
comprise cash on hand and amounts invested in underlying treasury and money market funds that are readily convertible to a known amount
of cash with three months or less from date of acquisition and are subject to an insignificant risk of change in value. As
of September 30, 2024 and March 31, 2024, cash equivalents was comprised of a money market account with maturities less than 90 days from
the date of purchase. Cash and cash equivalents are held with major international financial institutions and therefore the risk
of loss is minimal.
Liquidity Risk
Liquidity risk is the risk that the Company will encounter
difficulty in satisfying financial obligations as they become due.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 14. FINANCIAL INSTRUMENTS AND RISK MANAGEMENT (Cont'd)
The Company’s approach to managing liquidity is
to ensure, as far as possible, that it will have sufficient liquidity to meet its liabilities when due, under both normal and stressed
conditions without incurring unacceptable losses or risking harm to the Company’s reputation. The Company holds sufficient cash
and cash equivalents to satisfy current obligations under accounts payable and accruals.
The Company monitors its liquidity position regularly
to assess whether it has the funds necessary to meet its operating needs and needs for investing in new projects.
As a biotech company at an early stage of development
and without significant internally generated cash flows, there are inherent liquidity risks, including the possibility that additional
financing may not be available to the Company, or that actual drug development expenditures may exceed those planned. The current uncertainty
in global capital markets could have an impact on the Company’s future ability to access capital on terms that are acceptable to
the Company. There can be no assurance that required financing will be available to the Company. See Note 9, “Capital Stock and
Reserves,” for a discussion of the Company’s share offering.
Foreign Currency Risk
While the Company operates in various jurisdictions,
substantially all of the Company’s transactions are denominated in the U.S. Dollar, except the deferred tax liability in the U.K.
settleable in British pound sterling.
NOTE 15. CAPITAL MANAGEMENT
The Company considers the items included in shareholders’
equity as capital. The Company had accounts payable and accrued liabilities of approximately $0.8 million and lease liability - current
of $0.028 million as of September 30, 2024 (accounts payable and accrued liabilities of approximately $2.8 million as of March 31, 2024
and lease liability - current of $0.040 million as of March 31, 2024) and current assets of approximately $2.7 million as of September
30, 2024 (approximately $7.7 million as of March 31, 2024). The Company’s objectives when managing capital are to safeguard the
Company’s ability to continue as a going concern in order to pursue new business opportunities and to maintain a flexible capital
structure, which optimizes the costs of capital at an acceptable risk.
The Company manages the capital structure and makes adjustments
to it in light of changes in economic conditions and the risk characteristics of the underlying assets.
Nasdaq Listing Rule 5550(b)(1) requires companies listed
on the Nasdaq Capital Market to maintain shareholders’ equity of at least $2.5 million for continued listing. As of September 30,
2024, shareholders’ equity attributable to the owners of the Company was approximately $1.4 million (approximately $4.0 million
as of March 31, 2024). As a result, the Company is not currently in compliance with Nasdaq Listing Rule 5550(b)(1). Accordingly, the Company
expects Nasdaq to notify it of such non-compliance. The Company does not anticipate that the notification will have an immediate effect
on the listing of the Company’s ordinary shares on the Nasdaq Capital Market. In accordance with the Nasdaq Listing Rules, the Company
expects to have 45 calendar days from the date of the notification to submit a plan to regain compliance with Nasdaq Listing Rule 5550(b)(1).
If the Company’s compliance plan is accepted, it may be granted up to 180 calendar days from the date of the initial notification
to evidence compliance. If the compliance plan is not accepted or the Company is otherwise unable to provide evidence of compliance within
Nasdaq’s allotted timeframe, Nasdaq may take steps to delist the Company’s ordinary shares. There can be no assurance that
the Company will be able to increase its shareholders’ equity in the future.
The Company is not subject to any externally imposed
capital requirements and does not presently utilize any quantitative measures to monitor its capital. There have been no changes to the
Company’s approach to capital management during the six months ended September 30, 2024 and 2023.
PORTAGE BIOTECH INC.
Notes to Condensed Consolidated Interim Financial Statements
(U.S. Dollars)
(Unaudited – See Notice to Reader dated November 26,
2024)
NOTE 16. NON-CONTROLLING INTEREST
The details of non-controlling interest for the six months ended September
30, 2024 and 2023 are as follows (in thousands):
| Schedule of non-controlling interest | |
| | |
| |
| |
Saugatuck
and subsidiary | | |
| Non-controlling interest as of April 1, 2024 | |
$ | (693 | ) |
| Net loss attributable to non-controlling interest | |
| (10 | ) |
| Non-controlling interest as of September 30, 2024 | |
$ | (703 | ) |
| |
| |
Saugatuck
and subsidiary | | |
| Non-controlling interest as of April 1, 2023 | |
$ | (650 | ) |
| Net loss attributable to non-controlling interest | |
| (14 | ) |
| Non-controlling interest as of September 30, 2023 | |
$ | (664 | ) |
Saugatuck and subsidiary includes Saugatuck and its wholly-owned
subsidiary, Saugatuck Rx LLC.
Exhibit 99.2
PORTAGE BIOTECH INC.
THREE AND
SIX MONTHS ENDED SEPTEMBER 30, 2024
MANAGEMENT’S DISCUSSION AND
ANALYSIS
Prepared as of November 26, 2024
TABLE
OF CONTENTS
Management Discussion and
Analysis
The following
discussion and analysis by management of the financial condition and financial results for Portage Biotech Inc. for the three and six
months ended September 30, 2024, should be read in conjunction with the unaudited condensed consolidated interim financial statements
for the three and six months ended September 30, 2024, together with the related Management’s Discussion and Analysis and audited
consolidated financial statements for the year ended March 31, 2024, and the annual report on Form 20-F (our “Annual Report”)
for the fiscal year ended March 31, 2024 (“Fiscal 2024”).
Forward-Looking
Statements
This document
includes “forward-looking statements.” All statements, other than statements of historical facts, included herein or incorporated
by reference herein, including without limitation, statements regarding our business strategy, plans and objectives of management for
future operations and those statements preceded by, followed by or that otherwise include the words “believe,” “expects,”
“anticipates,” “intends,” “estimates,” “will,” “may,” “should,”
“could,” “targets,” “projects,” “predicts,” “plans,” “potential,”
or “continue,” or similar expressions or variations on such expressions are forward-looking statements. We can give no assurances
that such forward-looking statements will prove to be correct.
Each forward-looking
statement reflects our current view of future events and is subject to risks, uncertainties and other factors that could cause actual
results to differ materially from any results expressed or implied by our forward-looking statements.
We have
made the decision to discontinue our sponsored trial for the invariant natural killer T-cell (“iNKT”) program and pause further
patient accrual to our sponsored adenosine program for both PORT-6 and PORT-7. In the event that we resume these clinical trials and
further development of our programs, our risks and uncertainties include, but are not limited to:
|
• |
our plans and ability to develop and commercialize
product candidates and the timing of these development programs; |
|
• |
clinical development of our product candidates,
including the timing for availability and release of results of current and future clinical trials; |
|
• |
our expectations regarding regulatory communications,
submissions or approvals; |
|
• |
the potential functionality, capabilities, benefits
and risks of our product candidates as compared to others; |
|
• |
our maintenance and establishment of intellectual
property rights in our product candidates; |
|
• |
our need for financing and our estimates regarding
our capital requirements and future revenues and profitability; |
|
• |
our estimates of the size of the potential markets
for our product candidates; and |
|
• |
our selection and licensing of product candidates |
Our business
focus has been that of a pharmaceutical development business subject to all of the risks of a pharmaceutical development business. In
the event that we resume enrollment in the clinical trials and further development of our programs, we do not anticipate directly engaging
in the commercialization of the product candidates we develop.
These
statements are based on assumptions and analyses made by us in light of our experience and our perception of historical trends, current
conditions and expected future developments based on the focus of our business activities on biotechnology, as well as other factors
we believe are appropriate in particular circumstances. However, whether actual results and developments will meet our expectations and
predictions depends on a number of risks and uncertainties, which could cause actual results to differ materially from our expectations,
including the risks set forth under the heading “Business Environment – Risk Factors” below and in
Item 3 “Key Information – Risk Factors” in our Annual Report on Form 20-F for the year ended March 31, 2024.
Consequently,
all of the forward-looking statements made in this Management’s Discussion and Analysis are qualified by these cautionary statements.
We cannot assure you that the actual results or developments anticipated by us will be realized or, even if substantially realized, that
they will have the expected effect on us or our business or operations.
Unless the
context indicates otherwise the terms “Portage Biotech Inc.,” “the Company,” “our Company,” “Portage,”
“we,” “us” or “our” are used interchangeably in this Management’s Discussion and Analysis and
mean Portage Biotech Inc. and its subsidiaries. Capitalized terms used but not defined herein have the meaning ascribed to such terms
in our unaudited condensed consolidated interim financial statements for the three and six months
ended September 30, 2024.
Nature
of Operations and Overview
Due to our
future funding needs for clinical development of our programs as well as the current capital raising market for biotechnology companies,
we made the decision to discontinue the IMPORT-201 trial (PORT-2) and to pause further patient accrual to the ADPORT-601 trial (PORT-6
and PORT-7). The PORT-3 investigator trial is continuing, and all existing patients in the ADPORT-601 study will continue until disease
progression. We are continuing to collect and analyze data from these patients. We replaced a patient who withdrew and is unevaluable
for the 28-day dose limiting toxicity (“DLT”) period. We are exploring strategic alternatives, which may include finding
a partner for one or more of our assets, a sale of our company, a merger, restructurings, both in and out of court, company winddown,
further financing efforts or other strategic action. The following discussion reflects our operations in the event we are able to raise
additional capital to fund the clinical development of our programs.
We are a
clinical stage immuno-oncology company that advances treatments, in which we believe will be first-in-class therapies that target known
checkpoint resistant pathways, to improve long-term treatment response and the quality of life in patients with invasive cancers.
Our access
to next-generation technologies has provided us the capability to identify and understand biological mechanisms, clinical therapies and
product development strategies that have supported our programs through the translational pipeline and into the clinic.
We have
sourced and developed early- to mid-stage treatments that we believe will be first-in-class therapies for a variety of cancers, by funding,
implementing viable, cost-effective product development strategies, clinical counsel/trial design, shared services, financial and project
management to enable efficient, turnkey execution of commercially informed development plans. Our drug development pipeline portfolio
continues to encompass product candidates or technologies based on biology addressing known resistance pathways/mechanisms of current
checkpoint inhibitors with established scientific rationales.
Our Board
of Directors approved a reverse stock split of our ordinary shares at a ratio of 1-for-20. Beginning with the opening of trading on August
15, 2024, our ordinary shares began trading on Nasdaq on a split-adjusted basis under the existing trading symbol “PRTG”.
The reverse stock split was implemented to increase the per share trading price of our ordinary shares for the purpose of ensuring a
share price high enough to comply with the minimum $1.00 bid price requirement for continued listing on Nasdaq. We received notice from
Nasdaq on August 30, 2024 informing us that we had regained compliance with the minimum $1.00 bid price requirement for continued listing
on Nasdaq.
The Portage Approach
Our mission
has been to advance and grow a portfolio of innovative, early-stage oncology assets based on the latest scientific breakthroughs focused
on overcoming immune resistance and expanding the addressable patient population. Given these foundations, we have managed capital allocation
and risk as much as we have overseen drug development. By focusing our efforts on translational medicine and pipeline diversification,
we have sought to mitigate overall exposure to many of the inherent risks of drug development.
Our approach
has been guided by the following core elements:
|
• |
Portfolio diversification to mitigate risk and
maximize optionality; |
|
• |
Capital allocation based on risk-adjusted potential,
including staged funding to pre-specified scientific and clinical results; |
|
• |
Virtual infrastructure and key external relationships
to maintain a lean operating base; |
|
• |
Internal development capabilities complemented
by external business development; |
|
• |
Rigorous asset selection for broad targets with
disciplined ongoing evaluation; |
|
• |
Focus on translational medicine and therapeutic
candidates with single agent activity; |
|
• |
Conduct randomized trials early and test non-overlapping
mechanisms of action; and |
|
• |
Improve potential outcomes for patients with
invasive cancers. |
We have
executed such approach through our internal core team and our network of experts, contract labs and academic partners.
Our Science Strategy
Prior to
our decision to discontinue our iNKT IMPORT-201 sponsored trial and pause further patient accrual in the adenosine program, our goal
has been to develop immuno-oncology therapeutics that will dramatically improve the standard-of-care for patients with cancer. The key
elements of our scientific strategy have been to:
|
• |
Build a pipeline of differentiated oncology therapeutic
candidates that are diversified by mechanism, broad targets, therapeutic approach, modality, stage of development, leading to a variety
of deal types that can be executed with partners; |
|
• |
Expand our pipeline through research collaborations,
business development and internally designed programs; |
|
• |
Continue to advance and evolve our pipeline;
and |
|
• |
Evaluate strategic opportunities to accelerate
development timelines and maximize the value of our portfolio. |
Our Pipeline
We have
built a strong portfolio of immuno-oncology therapeutic candidates and programs that are diversified by mechanism, therapeutic approach,
modality and stage of development. Prior to our decision to discontinue further development of our iNKT sponsored trial and pause further
patient accrual to our sponsored adenosine program, we rigorously assessed each of our programs on an ongoing basis using internally
defined success criteria to justify continued investment and determine proper capital allocation. When certain programs do not meet our
de-risking criteria for advancement, we look to monetize or terminate those programs and preserve our capital and resources to invest
in programs with greater potential.
The charts
set forth below, illustrate the state of our immuno-oncology therapeutic product candidates and programs before development activities
were discontinued and/or paused. At this time PORT-3 is the only candidate currently actively recruiting and being evaluated as part
of an investigator sponsored study without funding from us. Additionally, notwithstanding our decision to pause further development of
PORT-2 and PORT-7 both INDs continue to remain active and the clinical trial for PORT-6 remains open, with further enrollment into additional
cohorts still on pause. We undertake no obligation to update or revise any forward-looking statements, whether as a result of new information,
future events or otherwise, except as required by law. Before you make an investment decision regarding us, you should make your own
analysis of forward-looking statements and our projections about candidate and program development and results.


Our Business Model
We are a
development organization that is structured to facilitate flexibility in financing and ease of partnering, licensing, and merger/acquisition
of individual assets and or technology platforms. The intellectual property (“IP”) for each platform is held in separate
private entities. Our employees and consultants work across the pipeline of assets and we believe that this can (i) enhance operational
efficiency, (ii) maintain an optimal cost structure, (iii) attract leading collaborators, and (iv) promote asset flexibility, as further
described below. If we were to resume enrollment in our clinical programs, we believe our experience and approach would continue to leverage
the operating and cost structures that are further described below.
|
• |
Enhance operational efficiency: We allocate
resources while empowering managers to make program-level decisions in order to increase productivity and speed. We believe this
model enables a flexible organizational structure that can achieve scale through the addition of programs without increasing burdensome
bureaucracy or redundant infrastructure. |
|
• |
Maintain an optimal cost structure: We
have a relatively small number of employees and have partnered with a number of service providers to leverage their infrastructure
and expertise as needed instead of embarking on capital-intensive lab, manufacturing, and equipment expenditures. By reducing overhead
costs, we believe we can increase the likelihood that we can generate a return on invested capital. |
|
• |
Attract leading collaborators and licensors:
Our pipeline is comprised of therapies we believe will be first-in-class therapies for a variety of cancers sourced via our industry
contacts and relationships (including academia and pharmaceutical industry executives). On preclinical programs/technology, we initially
established development structures enabling us to keep licensors economically incentivized at the program level. We believe that
our experienced drug development leadership team and approach to resource allocation differentiate us from other potential licensees. |
|
• |
Leverage the commoditized checkpoint marketplace
and explore the potential to further enhance long-term clinical benefits for patients with cancer and also expand the eligible population
to include those who do not currently receive anti-PD-1 therapy: Presently there are multiple approved checkpoint therapeutics
that lack differentiation, resulting in a competitive market dynamic, which will favor combination therapy. There remains opportunity
for potential expansion in the PD-1 market with our adenosine antagonists. Studies show that 70-80% of patients do not respond or
have a limited response to existing monotherapies, such as PD-1 checkpoint inhibitors. We see potential for our unique approach of
using adenosine antagonists to initiate an immune response in tumors that have become refractory to checkpoint therapy or to increase
the number of front-line patients achieving more durable responses. Combinations can improve this but often come at the cost of significant
additional toxicity. The market is saturated with at least 14 approved PD-1 antibodies, and every major pharmaceutical company competes
in this space. Extending the use of PD-1 antibodies could still provide a significant potential upside for companies competing for
market share. |
|
• |
Promote asset flexibility: Our structure
is designed to maximize flexibility and cost efficiency. This allows us to efficiently pursue various subsidiary-level transactions,
such as stock or asset sales, licensing transactions, strategic partnerships and/or co-development arrangements. It also provides
us with the flexibility to terminate programs with minimal costs if results do not meet our de-risking criteria for advancement. |
We are a
BVI business company incorporated under the BVI Business Companies Act (Revised Edition 2020, as amended) with our registered office
located at Clarence Thomas Building, P.O. Box 4649, Road Town, Tortola, British Virgin Islands, VG1110. Our U.S. agent, Portage Development
Services Inc., is located at 59 Wilton Road, Westport, CT 06880.
We
currently are a foreign private issuer under SEC rules. We are also a reporting issuer under the securities legislation of the provinces
of Ontario and British Columbia. Our ordinary shares were listed on the CSE under the symbol “PBT.U”. On February 25, 2021,
our ordinary shares began trading on the Nasdaq Capital Market under the symbol “PRTG”. As the principal market for our ordinary
shares is Nasdaq, we voluntarily delisted from the CSE on April 23, 2021.
Summary of Results
The following
table summarizes financial information for the quarter ended September 30, 2024, and the preceding eight quarters (in thousands except
net loss per share). All share and per share amounts reflect the 1-for-20 reverse stock split effected August 15, 2024.
| Quarter Ended |
|
|
Sept. 30,
2024 |
|
|
|
June 30,
2024 |
|
|
|
Mar.
31,
2024 |
|
|
|
Dec. 31,
2023 |
|
|
|
Sept. 30,
2023 |
|
|
|
June 30,
2023 |
|
|
|
Mar.
31,
2023 |
|
|
|
Dec. 31,
2022 |
|
|
|
Sept. 30
2022 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net loss attributable to owners of
the Company |
|
|
(1,360 |
) |
|
|
(1,656 |
) |
|
|
(24,889 |
) |
|
|
(39,373 |
) |
|
|
(5,158 |
) |
|
|
(5,919 |
) |
|
|
(94,448 |
) |
|
|
(7,485 |
) |
|
|
(949 |
) |
| Comprehensive loss attributable to the owners of the Company |
|
|
(1,360 |
) |
|
|
(1,656 |
) |
|
|
(28,371 |
) |
|
|
(36,398 |
) |
|
|
(6,458 |
) |
|
|
(4,150 |
) |
|
|
(95,714 |
) |
|
|
(11,502 |
) |
|
|
(949 |
) |
| Working capital (1) |
|
|
1,813 |
|
|
|
2,191 |
|
|
|
4,816 |
|
|
|
4,808 |
|
|
|
3,131 |
|
|
|
8,254 |
|
|
|
11,811 |
|
|
|
13,110 |
|
|
|
15,737 |
|
| Equity attributable to owners of the Company |
|
|
1,398 |
|
|
|
2,511 |
|
|
|
4,022 |
|
|
|
31,999 |
|
|
|
67,661 |
|
|
|
73,307 |
|
|
|
76,045 |
|
|
|
168,945 |
|
|
|
178,434 |
|
| Net loss per share - Basic |
|
|
(1.26 |
) |
|
|
(1.58 |
) |
|
|
(23.74 |
) |
|
|
(37.68 |
) |
|
|
(5.79 |
) |
|
|
(6.68 |
) |
|
|
(108.81 |
) |
|
|
(8.79 |
) |
|
|
(1.13 |
) |
| Net loss per share - Diluted |
|
|
(1.26 |
) |
|
|
(1.58 |
) |
|
|
(23.74 |
) |
|
|
(37.68 |
) |
|
|
(5.79 |
) |
|
|
(6.68 |
) |
|
|
(108.81 |
) |
|
|
(8.79 |
) |
|
|
(1.13 |
) |
|
(1) |
September
30, 2022 working capital is net of warrant liability of $8 settleable on a non-cash basis. |
Number
of Ordinary Shares
The following
table summarizes the number of our ordinary shares issued and outstanding at September 30, 2024 and November 26, 2024:
| |
|
|
September 30, 2024 |
|
|
|
November 26, 2024 |
|
| Shares issued (a) |
|
|
|
1,116,681 |
|
|
|
1,123,757 |
|
| Shares outstanding (a) |
|
|
|
1,116,249 |
|
|
|
1,121,059 |
|
|
(a) |
These
amounts exclude an aggregate of 12,150 restricted stock units granted to our executive chairman and an employee on January 13, 2021,
which vested immediately on the date of grant and are subject to certain restrictions for the settlement and delivery of the ordinary
shares underlying the restricted stock units and 5,165 restricted stock units granted to employees (one of whom is executive chairman)
on January 19, 2022, which vested immediately on the date of grant and are subject to certain restrictions for the settlement and
delivery of the ordinary shares underlying the restricted stock units. In January 2024, we issued 308 shares for the exercise of
470 RSUs net of 162 shares repurchased into treasury to pay for an employee’s payroll taxes. In September 2024, we issued 882
shares for the exercise of 1,152 RSUs net of 270 shares repurchased into treasury to pay for an employee’s payroll taxes. |
Business
Environment – Risk Factors
Please refer
to the Annual Report for Fiscal 2024 on Form 20-F as filed with the Securities and Exchange Commission
on August 14, 2024, for detailed information as the economic and industry factors are substantially unchanged as of the date hereof
other than as described below.
We
may not be able to regain, or maintain, compliance with the continued listing requirements of The Nasdaq Capital Market.
Our ordinary
shares are listed on the Nasdaq Capital Market, and we are therefore subject to its continued listing requirements, including requirements
with respect to the market value of our publicly-held shares, market value of our listed shares, minimum bid price per share, and minimum
shareholders’ equity, among others. If we fail to satisfy one or more of the requirements, we may be delisted from the Nasdaq Capital
Market.
Nasdaq Listing
Rule 5550(b)(1) requires companies listed on the Nasdaq Capital Market to maintain shareholders’ equity of at least $2.5 million
for continued listing. As of September 30, 2024, our shareholders’ equity was $1.398 million and, as a result, we are not currently
in compliance with Nasdaq Listing Rule 5550(b)(1). Accordingly, we expect Nasdaq to notify us of such non-compliance. We do not anticipate
that the notification will have an immediate effect on the listing of our ordinary shares on the Nasdaq Capital Market. In accordance
with the Nasdaq Listing Rules, we expect that we will have 45 calendar days from the date of the notification to submit a plan to regain
compliance with Nasdaq Listing Rule 5550(b)(1). If our compliance plan is accepted, we may be granted up to 180 calendar days from the
date of the initial notification to evidence compliance. If our compliance plan is not accepted or we are otherwise unable to evidence
compliance within Nasdaq’s allotted timeframe, Nasdaq may take steps to delist our ordinary shares. There can be no assurance that
we will be able to increase our shareholders’ equity in the future.
Delisting
from the Nasdaq Capital Market may adversely affect our ability to raise additional financing through the public or private sale of equity
securities, may significantly affect the ability of investors to trade our securities and may negatively affect the value and liquidity
of our ordinary shares. Delisting also could have other negative results, including the potential loss of employee confidence, the loss
of institutional investors or interest in business development opportunities.
If we are
delisted from Nasdaq and we are not able to list our ordinary shares on another exchange, our ordinary shares could be quoted on the
OTC Bulletin Board or in the “pink sheets.” As a result, we could face significant adverse consequences including, among
others:
|
• |
a limited availability of market quotations for
our securities; |
|
• |
a determination that our shares are a “penny
stock” which will require brokers trading in our ordinary shares to adhere to more stringent rules and possibly result in a
reduced level of trading activity in the secondary trading market for our securities; |
|
• |
a limited amount of news and little or no analyst
coverage for us; |
|
• |
we would no longer qualify for exemptions from
state securities registration requirements, which may require us to comply with applicable state securities laws; and |
|
• |
a decreased ability to issue additional securities
(including pursuant to short-form Registration Statements on Form F-3) due to the Baby Shelf Rule or obtain additional financing
in the future. |
Our
Programs and Technology – Recent Developments
After a
review of our future funding needs for clinical development of our programs as well as the current capital raising market for biotechnology
companies, we made the decision to discontinue our iNKT sponsored trial (the investigator sponsored trial of PORT-3 is ongoing without
financial support from us) and pause further patient accrual in our sponsored adenosine program. We are exploring strategic alternatives,
which may include finding a partner for one or more of our assets, a sale of our company, a merger, restructurings, both in and out of
court, a company wind down, further financing efforts or other strategic action. Below is a discussion of our clinical programs and the
status of such programs prior to our decision to discontinue our iNKT sponsored trial and pause further patient accrual to our sponsored
studies.
Adenosine Receptor Antagonist
Platform
A critical
mechanism of cancer immune evasion is the generation of high levels of immunosuppressive adenosine within the tumor microenvironment
(“TME”). Research suggests that the TME has significantly elevated concentrations of extracellular adenosine. Engagement
with adenosine receptors type 2A (“A2A”) and type 2B (“A2B”) triggers a dampening effect on the immune response,
suppressing effector cell function and stabilizing immunosuppressive regulatory cells. Over-expression of the A2A and A2B receptors leads
to a poor prognosis in multiple cancers, including prostate cancer, colorectal cancer and lung adenocarcinoma, driven by a reduced ability
to generate an immune response against the tumor.
These findings
have made A2A and A2B high-priority targets for immunotherapeutic intervention. Before pausing, we were advancing four adenosine antagonists
that we believe to be first-in-class, which together represent a broad suite of adenosine-targeting approaches and were expected to enable
a comprehensive exploration of how targeting the adenosine pathway could potentially improve response in multiple cancer and non-cancer
indications. By modulating the adenosine pathway in four different ways, we expected to determine the optimal approach to maximize the
impact of the mechanism of action on different tumors.
We have
designed the ADPORT-601 clinical trial to evaluate the activity and safety of PORT-6 and PORT-7 alone and in combination. If we resume
accrual, we would expect this trial to adapt over time and also include safety cohorts for these two agents with other immune activating
agents including others from our internal pipeline. Depending on the data, it can be expanded to evaluate either agent as monotherapy
or a randomized comparison of either agent plus standard of care versus standard of care alone.
PORT-6
(TT-10)
PORT-6 is
an A2A antagonist being studied for the treatment of A2A expressing solid tumors. We believe PORT-6 is more potent, more durable and
more selective than other clinical stage A2A agents.
Prior to
pausing patient enrollment in the clinical study, the ADPORT-601 Phase 1a trial for PORT-6 dosed its first patient in June 2023. There
have been a total of 12 patients dosed to date, with one patient continuing to receive treatment and the first two dose escalation cohorts
being completed. The third cohort has been fully enrolled, with only one patient experienced a serious adverse event (blurry vision and
stroke) that the investigator initially determined could possibly be related to PORT-6. With further follow-up, this event was classified
as unrelated to treatment. We remain encouraged by the two patients (discontinued) who received six or more lines of prior therapy and
experienced prolonged stable disease for more than eight months. After the study was put on hold, we made the decision to replace a patient
that withdrew treatment due to and unrelated adverse event prior to DLT assessment and that patient remains in the trial with stable
disease. Further recruitment remains on pause, with three investigator sites still open, while we explore strategic alternatives.
PORT-7
(TT-4)
PORT-7 is
an A2B antagonist for the treatment of solid tumors. PORT-7 has a very selective profile that focuses on A2B. PORT-7 is in Phase 1a from
an IND perspective, though we have not commenced dosing patients.
PORT-8
(TT-53)
PORT-8 is
a dual inhibitor of adenosine receptors 2A and 2B (A2A/A2B) to address solid tumors.
PORT-9
(TT-3)
PORT-9 is
an A2B antagonist designed to treat colorectal and gastrointestinal cancers. The PORT-9 program is a pre-clinical stage program.
In connection
with the adenosine program, we will focus on solid tumor types with high adenosine expression of receptors A2A and A2B and enrich for
patients that have high adenosine expression and therefore have potential to benefit most from treatment.
Other
Pipeline and Investee Programs
Prior to
our decision to discontinue our iNKT sponsored trial and pause further development of our adenosine program, we were focused on delivering
clinical data with the adenosine program described above and prioritizing the allocation of financial resources to that program. Developmental
work continued on some of the other developmental assets, through collaborations such as that with the U.S. National Cancer Institute
(“NCI”) and other academic groups, as further described below. These developmental assets may be re-evaluated at a future
point depending on market conditions, ongoing data, funding priorities and status.
Invariant Natural Killer
T-cells (iNKT cells) Platform
iNKT cells
play an important role in anti-tumor immune responses and are a distinct class of T lymphocyte displaying a limited diversity of T-cell
receptors. They recognize lipid antigens on the surface of tumor cells and produce large amounts of cytokines within hours of stimulation
without the need for clonal expansion. Furthermore, iNKT cells activate multiple immune system components, including dendritic cells
(“DC”), T-cells and B-cells and stimulate an antigen-specific expansion of these cells. Our operating subsidiary, iOx Therapeutics
Ltd. (“iOx”), holds an exclusive license (with the right to sub-license) from the Ludwig Institute for Cancer Research (the
“Ludwig Institute”) to use, research, develop and commercialize iNKT cell engagers, for the treatment of various forms of
human disease, including cancer, under the Ludwig Institute’s intellectual property and know-how.
PORT-2
(IMM60)
PORT-2 is
an iNKT cell engager formulated in a liposome with a six-member carbon head structure that has been shown to activate both human and
murine iNKT cells, resulting in DC maturation and the priming of Ag-specific T and B cells.
In animal
models, PORT-2 enhanced the frequency of tumor specific immune responses. iNKT cells are unique lymphocytes defined by their co-expression
of surface markers associated with NK cells along with a T-cell antigen receptor. They recognize amphipathic ligands such as glycolipids
or phospholipids presented in the context of the non-polymorphic, MHC class I-like molecule CD1d. Activated iNKT cells rapidly produce
IFN-gamma and IL-4 and induce DC maturation and IL-12 production.
In August
2021, we dosed the first patient in the IMP-MEL PORT-2 clinical trial, a Phase 1/2 dose escalation and randomized expansion trial. Prior
to discontinuing the PORT-2 trial, it was expected to enroll up to 88 patients with melanoma or non-small cell lung carcinoma (“NSCLC”)
in order to evaluate safety and efficacy. In November 2022, we announced that we had entered into a clinical trial collaboration with
Merck to evaluate PORT-2 in combination with pembrolizumab for patients with NSCLC. Under the terms of the collaboration, Merck supplied
pembrolizumab for our Phase 1/2 trial of PORT-2 in patients with NSCLC and melanoma. The trial was closed in June 2024. The Merck collaboration
terminated in December 2023.
Preliminary
Phase 1 data from the IMP-MEL PORT-2 clinical trial, presented at the Society for Immunotherapy of Cancer in November 2023, suggests
PORT-2 was well tolerated when administered as a monotherapy, with no related severe or serious adverse events. All possibly related
adverse events were mild to moderate and did not limit dosing. Given the favorable safety profile observed in the clinical trials to
date, the clinical protocol for the IMP-MEL PORT-2 clinical trial was amended to include a higher Phase 1 dose level as our near-term
focus was defining the recommended Phase 2 dose.
Prior to
our decision to discontinue further development of our iNKT platform, the combination safety cohort with pembrolizumab was being conducted
in parallel with the ongoing high dose monotherapy cohort. As of November 2023, two patients had received the combination with pembrolizumab,
and no related severe or serious adverse events were reported. The adverse event profile was consistent with pembrolizumab. Previously
reported biomarker data confirmed the mechanism of action (i.e., both activation of the innate and adaptive arms of the immune system).
The following figure illustrates the different lesion responses. Although these are preliminary results, several lesions showed shrinkage,
and the responses in liver metastases were encouraging.
Prior to
our decision to discontinue our iNKT sponsored trial, we were encouraged by the patient data set that we believe supports proof of concept
for using an iNKT engager in cancer treatment. Preliminary Phase 1 data suggests that PORT-2 has a favorable safety and tolerability
profile as a monotherapy at all doses tested to date (as noted above), has demonstrated evidence of single agent activity, and biomarkers
confirm mechanistic potential of PORT-2 to activate both the adaptive and innate immune systems.
The clinical
trial agreement has been transferred from the University of Oxford to us through our iOx subsidiary and the trial was converted to a
program sponsored by iOx.
Prior to
our decision to discontinue the iNKT sponsored trial, the protocol was being amended, given the safety data shown at the highest planned
doses, to escalate patient dosing to include one additional higher dose to identify the recommended Phase 2 dose. We are exploring strategic
alternatives, which may include finding a partner for one or more of our assets, a sale of our company, a merger, restructurings, both
in and out of court, a company wind down, further financing efforts or other strategic action.
PORT-3
(IMM65)
PORT-3 is
a poly(lactide-co-glycolide) (“PLGA”)-nanoparticle formulation of PORT-2 (IMM60) combined with a NY-ESO-1 peptide vaccine.
Biodegradable PLGA-nanoparticles function as a delivery platform for immunomodulators and tumor antigens to induce a specific anti-tumor
immune response. PLGA has minimal (systemic) toxicity and is used in various drug-carrying platforms as an encapsulating agent. Furthermore,
co-formulating an iNKT engager with a peptide vaccine in a particle has shown to be approximately five times more potent in killing cancer
cells and generating an antigen-specific CD8 T-cell response than giving the two agents individually.
NY-ESO-1
is a cancer-testis antigen expressed during embryogenesis and in the testis, an immune privileged site. Furthermore, NY-ESO-1 expression
is observed in several advanced cancers: Lung (2-32%), melanoma (40%), bladder (32-35%), prostate (38%), ovarian (30%), esophageal (24-33%),
and gastric cancers (8-12%). Clinical trials have shown the safety and tolerability of Good Manufacturing Practices-grade NY-ESO-1 peptides
in patients with cancer.
PORT-3 is
being evaluated as part of an investigator sponsored study without funding from us. The first patient was dosed in 2021 and patients
continue to enroll in the PRECIOUS Phase 1 trial of PORT-3 in patients with solid tumors. The Phase 1 portion of the trial is expected
to enroll 15 patients. The trial was having difficulty identifying tumors that expressed NY-ESO-1, so the trial protocol was amended
to include all solid tumors regardless of expression to facilitate assessment of safety. This platform is designed to demonstrate proof
of concept. The combination of NY-ESO-1 and IMM-60 is being evaluated to determine its ability to prime and boost an anti-tumor immune
response. Our patent position extends to other known tumor antigens, and, if we resume further development of our iNKT platform, we could
be prepared to rapidly launch other assets into the clinic if we see strong activity of this formulation. Preliminary safety data for
repeat dosing of PORT-3 in the PRECIOUS Phase 1 trial shows a favorable safety profile. The investigators with whom we work with have
continued to explore next generation targeted nanoparticles.
PORT-4, Nanolipogel (“NLG”)
co-formulation Platform
Scientists
are interested in novel ways to deliver multiple signals to the immune system in order to better activate an anti-tumor response. We
have been impressed with a platform from Yale University that allows different types of agents to be packaged together and will concentrate
them in tumors. We have licensed the platform for delivery of DNA aptamers and certain aptamer-small molecule-based combination products.
In order to have multiple proprietary agents with known mechanisms of action, we have licensed rights to create DNA aptamers for immune-oncology
targets and the first one developed is a proprietary PD1 aptamer, which has been placed in the NLG formulation. Early testing has shown
the formulation properly modulates PD1 signaling in vitro similar to a PD1 antibody I. In non-clinical, in vivo experiments, the NLG-PD1
performed favorably compared to a mouse PD1 antibody. We have conducted further research with the technology licensed from Yale University
to co-deliver a PD1 blocking signal with a small molecule vascular endothelial growth factor inhibitor.
As of September
30, 2024, we owned approximately 70% of the outstanding shares of Saugatuck Therapeutics, Ltd., the subsidiary on which the PORT-4 platform
is managed.
PORT-5, STING Agonist
Platform
Proprietary
immune priming and boosting technology (using a STING agonist delivered in a virus-like particle) has shown proof of concept in animal
models. This platform was developed to offer multiple ways to target immune stimulation towards the cancer, as well as to co-deliver
multiple signals in a single product. The PORT-5 STING platform’s advantage over chemical intratumoral approaches was potent immune
priming and boosting pathway within a virus-like particle to enable convenient systemic administration and traffic to the correct targets.
This technology would target dendritic cells, which is differentiated from other chemical STING approaches. To that end, Stimunity S.A.
(“Stimunity”) received grant funding to study this technology with any COVID-19 vaccine to evaluate if it is possible to
boost the immune response for immunocompromised or elderly patients. During April 2022, the American Association for Cancer Research
showcased PORT-5 preclinical data at a late-breaking session that shows that one or more targeted immunotherapy agents could be packaged
within a virus-like particle to increase potency, while enabling a selective immune activation. Stimunity was unable to raise any outside
funding, and activities were scaled back due to our own liquidity issues.
In December
2023, we completed a transfer of our equity in Stimunity and the Stimunity Convertible Note to iOx. In connection with that transfer,
the Stimunity Convertible Note was converted into 1,768 Class A shares of Stimunity.
As of September
30, 2024, we owned approximately 48.9% of the outstanding shares of Stimunity, the subsidiary on which the PORT-5 platform is managed.
We have made the decision not to further fund Stimunity’s operations and wrote down the remaining balance of our investment of
$1.0 million to nil as of March 31, 2024.
Early-Stage Research and
Development Collaborations
We have
also been interested in evaluating and testing new antibody targets in the suppressive tumor microenvironment with the goal to down regulate
or remove MDSC, TAMs, Tregs and other signals that impede the immune response from clearing cancer cells.
|
• |
We continue to collaborate with Dr. Robert Negrin
and his team at Stanford University in an investigator sponsored trial (“IST”) study to evaluate the use of PORT-2 with
iNKT cell therapies in animals. This work was intended to evaluate if an engager co-administered with expanded or transformed iNKT
cells can further activate the transplanted and endogenous cells inside the patient. The Stanford collaboration was also expected
to study the impact iNKT engagers have on driving an adaptive immune response and correcting the suppressive tumor microenvironment.
This IST remains operational as of the date of this report. |
|
• |
We entered into a Cooperative Research and Development
Agreement (“CRADA”) with the NCI. We and NCI planned to advance preclinical and potential clinical development of STING
agonists and anti-RAGE agents for cancer vaccines. After the acquisition of Tarus Therapeutics, LLC (“Tarus”), we amended
the CRADA to include exploration of the different adenosine compounds. We did not extend the CRADA beyond its current term and made
a termination payment of $62,500 in June 2024 as required under the contract. |
|
• |
We have a collaboration with Dr. Carmela de Santos
at University of Birmingham for the use of iNKT agents to treat sarcomas. Dr. de Santos has tested PORT-2 in human sarcoma cell lines
and has grant funding to test it in animal models. |
|
• |
We have a collaboration to study the use of adenosine
2A and adenosine 2B agents in mesothelioma with Drs. Luciano Mutti from Sbarro Institute for Cancer Research and Molecular Medicine,
Department of Biology, College of Science and Technology, Temple University and Dr. Steven Gray of St. James Hospital in Dublin. |
|
• |
There are other collaborations with experts in
the products areas for which we provide access to our compounds and collaborates on studies. |
Three Months Ended September
30, 2024 Compared to the Three Months Ended September 30, 2023
Results
of Operations
The following
details major expenses for the three months ended September 30, 2024, compared to the three months ended September
30, 2023 (in thousands):
| Three months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Operating expenses |
|
$ |
(1,604 |
) |
|
$ |
(5,930 |
) |
| Change in fair value of warrant liability |
|
|
(716 |
) |
|
|
— |
|
| Change in fair value of deferred purchase price payable – Tarus and deferred
obligation – iOx milestone |
|
|
— |
|
|
|
(113 |
) |
| Gain on settlement with Parexel – iOx CRO |
|
|
946 |
|
|
|
— |
|
| Share of loss in associate accounted for using equity method |
|
|
— |
|
|
|
(40 |
) |
| Depreciation expense |
|
|
(7 |
) |
|
|
(15 |
) |
| Foreign exchange transaction loss |
|
|
(5 |
) |
|
|
(17 |
) |
| Interest income, net |
|
|
24 |
|
|
|
43 |
|
| Loss before provision for income taxes |
|
|
(1,362 |
) |
|
|
(6,072 |
) |
| Income tax benefit |
|
|
— |
|
|
|
907 |
|
| Net loss |
|
|
(1,362 |
) |
|
|
(5,165 |
) |
| Other comprehensive loss |
|
|
|
|
|
|
|
|
| Net unrealized loss on investments |
|
|
— |
|
|
|
(1,300 |
) |
| Total comprehensive loss for period |
|
$ |
(1,362 |
) |
|
$ |
(6,465 |
) |
| |
|
|
|
|
|
|
|
|
| Comprehensive loss attributable to: |
|
|
|
|
|
|
|
|
| Owners of the Company |
|
$ |
(1,360 |
) |
|
$ |
(6,458 |
) |
| Non-controlling interest |
|
|
(2 |
) |
|
|
(7 |
) |
| Total comprehensive loss for period |
|
$ |
(1,362 |
) |
|
$ |
(6,465 |
) |
Results of Operations
for the Three Months Ended September 30, 2024 Compared to the Three Months Ended September 30, 2023
We incurred
a net loss of approximately $1.4 million during the three months ended September 30, 2024 (the “Fiscal 2025 Quarter”), compared
to a net loss of approximately $5.2 million during the three months ended September 30, 2023 (the “Fiscal 2024 Quarter”),
a decrease in net loss of $3.8 million, quarter-over-quarter.
The components
of the change in net loss and total comprehensive loss are as follows:
|
• |
Operating expenses, which include research and
development (“R&D”) costs and general and administrative (“G&A”) expenses, were $1.6 million in the
Fiscal 2025 Quarter, compared to $5.9 million in the Fiscal 2024 Quarter, a decrease of $4.3 million, which is discussed more fully
below. |
|
• |
A net gain of $0.9 million from the settlement
and release of obligations and liabilities under the Master Services Agreement between iOx and Parexel International (IRL) Limited
(“Parexel”). See “iOx – Parexel Master Services Agreement” section of Note 12 “Commitments and
Contingent Liabilities” for additional discussion regarding this matter included elsewhere in this Report. |
|
• |
A non-cash loss totaling $0.113 million comprised
of the change (increase) in the fair value of the deferred obligation - iOx milestone of $0.028 million, and the change (increase)
in the fair value of the deferred purchase price payable to the former Tarus shareholders of $0.085 million in the Fiscal 2024 Quarter. |
|
• |
A $0.7 million non-cash loss
from the change in the fair value of certain warrants accounted for as liabilities issued in connection with the
Private Placement (defined below) in October 2023. |
|
• |
Additionally, we reflected a marginal income
tax expense in the Fiscal 2025 Quarter, compared to a net deferred income tax benefit of $0.9 million in the Fiscal 2024 Quarter.
For the Fiscal 2024 Quarter, we recognized a decrease in net deferred tax liability of $0.3
million to reflect the effect of the change in exchange rates on the liability during the period and the recognition of $0.6 million
of current period losses in the U.K. |
Total comprehensive
loss in the Fiscal 2024 Quarter includes $1.3 million unrealized loss on investments compared
to nil in the Fiscal 2025 Quarter. The difference between net loss and total comprehensive loss in the Fiscal 2024 Quarter was due to
the effect of the unrealized non-cash loss with respect to our investment in Intensity Therapeutics, Inc. (“Intensity”).
We recognized a non-cash unrealized loss of $1.3 million in the Fiscal 2024 Quarter, representing the change in the fair value of the
investment in Intensity based on the Nasdaq listed price of the shares during the period.
Operating Expenses
Total operating
expenses are comprised of the following (in thousands):
| Three months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Research and development |
|
$ |
723 |
|
|
$ |
4,237 |
|
| General and administrative expenses |
|
|
881 |
|
|
|
1,693 |
|
| Total operating expenses |
|
$ |
1,604 |
|
|
$ |
5,930 |
|
Research and Development
Costs
R&D
costs are comprised of the following (in thousands):
| Three months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Research and development – Clinical |
|
$ |
366 |
|
|
$ |
1,962 |
|
| Payroll-related expenses |
|
|
245 |
|
|
|
367 |
|
| Consulting fees |
|
|
32 |
|
|
|
247 |
|
| Manufacturing costs |
|
|
29 |
|
|
|
968 |
|
| Research and development – CRADA |
|
|
— |
|
|
|
31 |
|
| Licensing fees |
|
|
— |
|
|
|
120 |
|
| Legal regarding patents’ registration |
|
|
5 |
|
|
|
— |
|
| Research and development services and storage |
|
|
46 |
|
|
|
148 |
|
| Share-based compensation expense |
|
|
— |
|
|
|
394 |
|
| Total research and development costs |
|
$ |
723 |
|
|
$ |
4,237 |
|
R&D
costs decreased by approximately $3.5 million, or approximately 83%, from approximately $4.2 million in the Fiscal 2024 Quarter to approximately
$0.7 million in the Fiscal 2025 Quarter. The decrease was primarily attributable to the winding down of clinical trial costs (principally
CRO-related), which decreased by approximately $1.6 million, from $2.0 million in the Fiscal 2024 Quarter to $0.4 million in the Fiscal
2025 Quarter, as activities ramped down throughout the period since we made the decision to pause enrollment in our sponsored clinical
trials in the third and fourth quarters of Fiscal 2024. Manufacturing-related costs decreased by $0.9 million, from $1.0 million in the
Fiscal 2024 Quarter to $0.029 million in the Fiscal 2025 Quarter. These decreases reflect the winding down of clinical activity and manufacturing-related
costs resulting from our decision to discontinue our sponsored clinical trial for the iNKT program and pause further patient accrual
to our sponsored adenosine program. R&D non-cash share-based compensation expense decreased from $0.4 million in the Fiscal 2024
Quarter to nil in the Fiscal 2025 Quarter. Payroll-related expenses decreased by $0.1 million, from $0.37 million in the Fiscal 2024
Quarter to $0.24 million in the Fiscal 2025 Quarter, due to the resignation of two employees in January 2024. Additionally, consulting
fees decreased by approximately $0.2 million from $0.25 million in the Fiscal 2024 Quarter
to $0.03 million in the Fiscal 2025 Quarter, to reflect the decrease in activity period-over-period. Finally, licensing fees decreased
by approximately $0.1 million due to licensing fees paid to
the licensor of certain intellectual property utilized in the iNKT clinical trial
in Fiscal 2024 Quarter compared to nil in Fiscal 2025 Quarter as the iNKT clinical trial was discontinued in the latter half of Fiscal
2024.
General and Administrative
Expenses
Key components
of G&A expenses are the following (in thousands):
| Three months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Professional fees |
|
$ |
398 |
|
|
$ |
762 |
|
| Payroll-related expenses |
|
|
134 |
|
|
|
223 |
|
| D&O insurance |
|
|
160 |
|
|
|
175 |
|
| Office and general expenses |
|
|
33 |
|
|
|
99 |
|
| Directors’ fees |
|
|
— |
|
|
|
83 |
|
| Share-based compensation expense |
|
|
141 |
|
|
|
348 |
|
| Consulting fees |
|
|
15 |
|
|
|
3 |
|
| Total general and administrative expenses |
|
$ |
881 |
|
|
$ |
1,693 |
|
G&A
expenses decreased by approximately $0.8 million, or approximately 48%, from approximately $1.7 million in the Fiscal 2024 Quarter to
approximately $0.9 million in the Fiscal 2025 Quarter. Professional fees decreased by $0.4 million, from $0.8 million in the Fiscal 2024
Quarter to $0.4 million in the Fiscal 2025 Quarter. Payroll-related expenses decreased by $0.1 million from $0.2 million in the Fiscal
2024 Quarter to $0.1 million in the Fiscal 2025 Quarter. The decrease in professional fees and payroll-related expenses is due to the
accrual of the monthly fees and payments for the entire second quarter in the first quarter for a consultant and employee in connection
with the Retention Agreements entered into on July 22, 2024. For further discussion regarding this matter, see “Retention Agreements
and General Releases” section of Note 13 “Related Party Transactions” included elsewhere in this Report. Additionally,
G&A non-cash share-based compensation expense decreased by $0.2 million due to the continued vesting of stock options, partially
offset by recording all share-based compensation expense as G&A expenses as the result of the discontinuation of the iNKT trial and
the pause of further patient accrual in the adenosine program. Additionally, directors’ fees decreased by $0.1 million in the Fiscal
2025 Quarter, compared to the Fiscal 2024 Quarter, as all directors, except for two directors who resigned in April 2024, waived their
fees in the Fiscal 2025 Quarter.
Six Months Ended September
30, 2024 Compared to the Six Months Ended September 30, 2023
Results of Operations
The following
details major expenses for the six months ended September 30, 2024, compared to the six months ended September
30, 2023 (in thousands):
| Six months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Operating expenses |
|
$ |
(4,443 |
) |
|
$ |
(10,927 |
) |
| Change in fair value of warrant liability |
|
|
426 |
|
|
|
— |
|
| Change in fair value of deferred purchase price payable – Tarus and deferred
obligation – iOx milestone |
|
|
— |
|
|
|
(1,224 |
) |
| Gain on settlement with Parexel – iOx CRO |
|
|
946 |
|
|
|
— |
|
| Share of loss in associate accounted for using equity method |
|
|
— |
|
|
|
(90 |
) |
| Foreign exchange transaction (loss) gain |
|
|
(7 |
) |
|
|
1 |
|
| Depreciation expense |
|
|
(15 |
) |
|
|
(26 |
) |
| Interest income, net |
|
|
69 |
|
|
|
123 |
|
| Loss before provision for income taxes |
|
|
(3,024 |
) |
|
|
(12,143 |
) |
| Income tax (expense) benefit |
|
|
(2 |
) |
|
|
1,052 |
|
| Net loss |
|
|
(3,026 |
) |
|
|
(11,091 |
) |
| Other comprehensive loss |
|
|
|
|
|
|
|
|
| Net unrealized gain on investments |
|
|
— |
|
|
|
469 |
|
| Total comprehensive loss for period |
|
$ |
(3,026 |
) |
|
$ |
(10,622 |
) |
|
|
|
|
|
|
|
|
|
| Comprehensive loss attributable to: |
|
|
|
|
|
|
|
|
| Owners of the Company |
|
$ |
(3,016 |
) |
|
$ |
(10,608 |
) |
| Non-controlling interest |
|
|
(10 |
) |
|
|
(14 |
) |
| Total comprehensive loss for period |
|
$ |
(3,026 |
) |
|
$ |
(10,622 |
) |
Results of Operations
for the Six Months Ended September 30, 2024 Compared to the Six Months Ended September 30,
2023
We incurred
a net loss of approximately $3.0 million during the six months ended September 30, 2024 (the “Fiscal 2025 Six
Months”), compared to net loss of $11.1 million during the six months ended September
30, 2023 (the “Fiscal 2024 Six Months”) and total comprehensive loss of approximately $3.0 million and $10.6 million during
the Fiscal 2025 Six Months and the Fiscal 2024 Six Months, respectively, a decrease in net loss of $8.1 million and a decrease
in comprehensive loss of $7.6 million, period-over-period.
The components
of the change in net loss and total comprehensive loss are as follows:
|
• |
Operating expenses, which include R&D and
G&A expenses, were $4.4 million in the Fiscal 2025 Six Months, compared to $10.9
million in the Fiscal 2024 Six Months, a decrease of $6.5 million, which is discussed more fully below. |
|
• |
A net gain of $0.9 million from the settlement
and release of obligations and liabilities under the Master Services Agreement between iOx and Parexel. See “iOx – Parexel
Master Services Agreement” section of Note 12 “Commitments and Contingent Liabilities” for additional discussion
regarding this matter included elsewhere in this Report. |
|
• |
A $0.4 million non-cash gain from the change
in the fair value of certain warrants accounted for as liabilities issued in connection with the Private Placement in October 2023. |
|
• |
A non-cash loss totaling $1.2 million comprised
of the change (increase) in the fair value of the deferred obligation - iOx milestone of $0.45 million, and the change (increase)
in the fair value of the deferred purchase price payable to the former Tarus shareholders of $0.77 million in the Fiscal 2024 Six
Months. |
|
• |
Additionally, we reflected a marginal income
tax expense in the Fiscal 2025 Six Months, compared to a net deferred income tax benefit of $1.1 million benefit
in the Fiscal 2024 Six Months. For the Fiscal 2024 Six Months, we recognized a decrease in net deferred tax liability of $0.1
million to reflect the effect of the change in exchange rates on the liability during the period and the recognition $1.0 million
of current period losses in the U.K. |
Total comprehensive
loss in the Fiscal 2024 Six Months includes $0.5 million unrealized gain on investments
compared to nil in the Fiscal 2025 Six Months. The difference between net loss and total comprehensive loss in the Fiscal 2024 Six Months
was due to the effect of the unrealized non-cash gain with respect to our investment in Intensity. We recognized a non-cash unrealized
gain of $0.5 million in the Fiscal 2024 Six Months, representing the change in the fair value of the investment in Intensity based on
the Nasdaq listed price of the shares during the period.
Operating Expenses
Total operating
expenses are comprised of the following (in thousands):
| Six months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Research and development |
|
$ |
2,028 |
|
|
$ |
7,865 |
|
| General and administrative expenses |
|
|
2,415 |
|
|
|
3,062 |
|
| Total operating expenses |
|
$ |
4,443 |
|
|
$ |
10,927 |
|
Research and Development Costs
R&D
costs are comprised of the following (in thousands):
| Six months ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Research and development – Clinical |
|
$ |
1,072 |
|
|
$ |
2,985 |
|
| Payroll-related expenses |
|
|
494 |
|
|
|
847 |
|
| Consulting fees |
|
|
153 |
|
|
|
460 |
|
| Manufacturing costs |
|
|
123 |
|
|
|
1,715 |
|
| Research and development – CRADA |
|
|
62 |
|
|
|
63 |
|
| Licensing fees |
|
|
38 |
|
|
|
232 |
|
| Legal regarding patents’ registration |
|
|
28 |
|
|
|
8 |
|
| Research and development services and storage |
|
|
58 |
|
|
|
238 |
|
| Share-based compensation expense |
|
|
— |
|
|
|
817 |
|
| Contractual milestone |
|
|
— |
|
|
|
500 |
|
| Total research and development costs |
|
$ |
2,028 |
|
|
$ |
7,865 |
|
R&D
costs decreased by approximately $5.8 million, or approximately 74%, from approximately $7.9 million in the Fiscal 2024 Six Months to
approximately $2.0 million in the Fiscal 2025 Six Months. The decrease was primarily attributable to the winding down of clinical trial
costs (principally CRO-related), which decreased by approximately $2.0 million, from $3.0 million in the Fiscal 2024 Six Months to $1.0
million in the Fiscal 2025 Six Months, as activities ramped down throughout the period since we made the decision to pause enrollment
in our sponsored clinical trials in the third and fourth quarters of Fiscal 2024. Manufacturing-related costs decreased by $1.6 million,
from $1.7 million in the Fiscal 2024 Six Months to $0.1 million in the Fiscal 2025 Six Months. These decreases reflect the winding down
of clinical activity and manufacturing-related costs resulting from our decision to discontinue our sponsored clinical trial for the
iNKT program and pause further patient accrual to our sponsored adenosine program. R&D non-cash share-based compensation expense
decreased from $0.8 million in the Fiscal 2024 Six Months to nil in the Fiscal 2025 Six Months. Payroll-related expenses decreased by
$0.35 million, from $0.85 million in the Fiscal 2024 Six Months to $0.5 million in the Fiscal 2025 Six Months, due to the resignation
of two employees in January 2024. Additionally, in the Fiscal 2024 Six Months, we incurred a milestone payment of $0.5 million for dosing
our first adenosine patients, a decrease in consulting fees of approximately $0.3 million from $0.5 million in the Fiscal 2024 Six
Months to $0.2 million in the Fiscal 2025 Six Months, to reflect the decrease in activity period-over-period, and, finally, a
decrease of $0.2 million in licensing fees paid to the licensor of certain intellectual property utilized in the iNKT clinical trial
prior to discontinuing the study in the latter half of Fiscal 2024.
General and Administrative Expenses
Key components of G&A expenses are the following
(in thousands):
| Six months
ended September 30, |
|
|
2024 |
|
|
|
2023 |
|
| |
|
|
|
|
| Professional fees |
|
$ |
941 |
|
|
$ |
1,232 |
|
| Payroll-related expenses |
|
|
771 |
|
|
|
447 |
|
| D&O insurance |
|
|
320 |
|
|
|
350 |
|
| Office and general expenses |
|
|
76 |
|
|
|
168 |
|
| Directors’ fees |
|
|
7 |
|
|
|
165 |
|
| Share-based compensation expense |
|
|
285 |
|
|
|
695 |
|
| Consulting fees |
|
|
15 |
|
|
|
5 |
|
| Total general and administrative
expenses |
|
$ |
2,415 |
|
|
$ |
3,062 |
|
G&A
expenses decreased by approximately $0.647 million, or approximately 21%, from approximately $3.062 million in the Fiscal 2024 Six Months
to approximately $2.415 million in the Fiscal 2025 Six Months. Professional fees decreased by $0.3 million, from $1.2 million in the
Fiscal 2024 Six Months to $0.9 million in the Fiscal 2025 Six Months due to decreased accounting and public relations related fees. Payroll-related
expenses increased by $0.4 million from $0.4 million in the Fiscal 2024 Six Months to $0.8 million in the Fiscal 2025 Six Months due
to the amounts associated with retention agreements executed with an employee and a consultant. For further discussion regarding this
matter, see “Retention Agreements and General Releases” section of Note 13 “Related Party Transactions” included
elsewhere in this Report. Additionally, G&A non-cash share-based compensation expense decreased by $0.4 million due to the continued
vesting of stock options, partially offset by recording all share-based compensation expense as G&A expenses as the result of the
discontinuation of the iNKT trial and the pause of further patient accrual in the adenosine program. Additionally, directors’ fees
decreased by $0.2 million in the Fiscal 2025 Six Months, compared to the Fiscal 2024 Six Months, as all directors, except for two directors
who resigned in April 2024, waived their fees in the Fiscal 2025 Six Months.
Liquidity
and Capital Resources
Capital Resources
We filed
the March 2021 Registration Statement with the SEC in order to sell ordinary shares, debt securities, warrants and units in one or more
offerings from time to time, which became effective on March 8, 2021. In connection with the March 2021 Registration Statement, we have
filed with the SEC:
|
• |
a base prospectus, which covers the offering,
issuance and sale by us of up to $200 million in the aggregate of the securities identified above from time to time in one or more
offerings; |
|
• |
a prospectus supplement, which covers the offer,
issuance and sale by us in an ATM offering program of up to a maximum aggregate offering price of $50 million of our ordinary shares
that may be issued and sold from time to time under the Sales Agreement with Cantor Fitzgerald & Co., the sales agent; |
|
• |
a prospectus supplement dated June 24, 2021,
for the offer, issuance and sale by us of 57,500 ordinary shares for gross proceeds of approximately
$26.5 million in a firm commitment underwritten public offering with Cantor Fitzgerald; |
|
• |
a prospectus supplement dated August 19, 2022,
for the resale of up to $30 million in ordinary shares that we may sell from time to time to Lincoln and an additional 4,726 shares
that were issued to Lincoln; and |
|
• |
a prospectus supplement dated September 29, 2023
for the offer, issuance and sale by us in a registered direct public offering through H.C. Wainwright & Co., the placement agent,
to an institutional and accredited investor of (i) 98,500 of our ordinary shares at a purchase price of $38.00 per share and (ii)
Pre-Funded Warrants to purchase up to 59,395 of our ordinary shares, at a purchase price of $37.98 per Pre-Funded Warrant), for aggregate
gross proceeds of approximately $6 million (the “2023 Equity Financing”). All Pre-Funded Warrants, which were exercisable
for one ordinary share at an exercise price of $0.02 per share, were exercised in full on May 29, 2024. |
The Sales
Agreement permits us to sell in an ATM program up to $50 million of ordinary shares from time to time. Through September 30, 2024, we raised approximately $4.2 million in gross proceeds through the sale of shares of common shares under the
ATM program. The sales under the prospectus
will be deemed to be made pursuant to an ATM program as defined in Rule 415(a)(4) promulgated under the Securities Act of 1933, as amended
(the “Securities Act”).
During
Fiscal 2024, we sold 9,331 ordinary shares under the ATM program, generating net proceeds of approximately $0.7 million. There were no shares sold under the ATM program during the six months ended September 30, 2024.
The March
2021 Registration Statement expired on March 8, 2024. In order to issue additional shares under our ATM program or the Committed Purchase
Agreement in the future, we would be required to file a new registration statement, which must be declared effective by the SEC prior
to use, and to file a prospectus supplement related to the ATM program or the Committed Purchase Agreement, as the case may be.
Furthermore,
our ATM program and the Committed Purchase Agreement with Lincoln are generally limited based on, among other things, our Nasdaq trading
volume. Under the Baby Shelf Rule, the amount of funds we can raise through primary public offerings of securities in any 12-month period
using a registration statement on Form F-3 is limited to one-third of the aggregate market value of the ordinary shares held by our non-affiliates,
which limitation may change over time based on our stock price, number of ordinary shares outstanding and the percentage of ordinary
shares held by non-affiliates. We are therefore limited by the Baby Shelf Rule as of the filing of this Form 6-K, until such time as
our non-affiliate public float exceeds $75 million.
In connection
with the 2023 Equity Financing, on September 29, 2023, we entered into the Purchase Agreement with an institutional and accredited investor
in connection with the Registered Direct Offering and the concurrent private placement. The Offerings closed on October 3, 2023.
Pursuant
to the Purchase Agreement, in the Registered Direct Offering, we sold (i) 98,500 of our ordinary shares, at a purchase price of $38.00
per share and (ii) Pre-Funded Warrants to purchase up to 59,395 Pre-Funded Warrant Shares. All Pre-Funded Warrants, which were immediately
exercisable for one ordinary share at an exercise price of $0.02 per share, were exercised in full on May 29, 2024.
In the Private
Placement, we issued to such institutional and accredited investor unregistered Series A Warrants to purchase up to 157,895 ordinary
shares, unregistered Series B Warrants to purchase up to 157,895 ordinary shares, and unregistered Series C Warrants to purchase up to
157,895 ordinary shares, together exercisable for an aggregate of up to 473,685 Private Warrant Shares. Pursuant to the terms of the
Purchase Agreement, for each ordinary share and Pre-Funded Warrant issued in the Registered Direct Offering, an accompanying Series A
Warrant, Series B Warrant and Series C Warrant were issued to such institutional and accredited investor. Each Series A Warrant is exercisable
for one Private Warrant Share at an exercise price of $38.00 per share, is immediately exercisable and will expire 18 months from the
date of issuance. Each Series B Warrant is exercisable for one Private Warrant Share at an exercise price of $45.20 per share, is immediately
exercisable and will expire three years from the date of issuance. Each Series C Warrant is exercisable for one Private Warrant Share
at an exercise price of $45.20 per share, is immediately exercisable and will expire five years from the date of issuance. The net proceeds
to us from the Offerings were approximately $5.3 million, after deducting placement agent’s fees and estimated offering expenses.
Pursuant
to an engagement letter, dated as of August 26, 2023, between us and H.C. Wainwright & Co., LLC (the “Placement Agent”),
we paid the Placement Agent a total cash fee equal to 6.0% of the aggregate gross proceeds received in the Offerings, or $0.36 million.
We also agreed to pay the Placement Agent in connection with the Offerings a management fee equal to 1.0% of the aggregate gross proceeds
raised in the Offerings ($0.06 million), $75,000 for non-accountable expenses and $15,950 for clearing fees. In addition, we agreed to
issue to the Placement Agent, or its designees, Placement Agent Warrants to purchase up to 7,896 ordinary shares, which represents 5.0%
of the aggregate number of ordinary shares and Pre-Funded Warrants sold in the Registered Direct Offering. The Placement Agent Warrants
have substantially the same terms as the Private Warrants, except that the Placement Agent Warrants have an exercise price equal to $47.50,
or 125% of the offering price per ordinary share sold in the Registered Direct Offering, and will be exercisable for five years from
the commencement of the sales pursuant to the Offerings.
Going Concern
The accompanying
condensed consolidated interim financial statements for the three and six months ended September 30, 2024 have been prepared on a basis
that assumes that we will continue as a going concern and that contemplates the continuity of operations, the realization of assets and
the satisfaction of liabilities and commitments in the normal course of business. Accordingly, the accompanying condensed consolidated
interim financial statements for the three and six months ended September 30, 2024 do not include any adjustments relating to the recoverability
and classification of recorded asset amounts or amounts of liabilities that might result from the outcome of this uncertainty.
As of September
30, 2024, we had cash and cash equivalents of approximately $1.8 million and total current liabilities of approximately $0.9 million.
For the six months ended September 30, 2024, we are reporting a net loss of approximately $3.0 million, and cash used in operating activities
of approximately $3.3 million. As of November 25, 2024, we had approximately $1.7 million of cash and cash equivalents on hand.
In late
Fiscal 2024, because of continued liquidity constraints, we made the decision to discontinue our iNKT sponsored trial and pause further
patient accrual to our adenosine program. We are exploring strategic alternatives, which may include finding a partner for one or more
of our assets, a sale of our company, a merger, restructurings, both in and out of court, a company wind down, further financing efforts
or other strategic action.
There can
be no assurance that our evaluation of strategic alternatives will result in any agreements or transactions, or that, if completed, any
agreements or transactions will be successful or on attractive terms. Any potential transaction would be dependent on a number of factors
that may be beyond our control, including, among other things, market conditions, industry trends, the interest of third parties in a
potential transaction with us and the availability of financing to us or third parties in a potential transaction with us on reasonable
terms. The process of reviewing strategic alternatives may require us to incur additional costs and expenses. It could negatively impact
our ability to attract, retain and motivate key employees, and expose us to potential litigation in connection with this process or any
resulting transaction. If we are unable to effectively manage the process, our financial condition and results of operations could be
adversely affected. In addition, any strategic alternative that may be pursued and completed ultimately may not deliver the anticipated
benefits or enhance shareholder value. There can be no guarantee that the process of evaluating strategic alternatives will result in
our company entering into or completing a potential transaction within the anticipated timing or at all. There is no set timetable for
this evaluation and we do not intend to disclose developments with respect to this evaluation unless and until we determine that further
disclosure is appropriate or legally required. As of November 25, 2024, we had approximately $1.7 million of cash and cash equivalents
on hand, which we expect is only sufficient to cover our operating needs through January 2025. These factors raise substantial doubt
about our ability to continue as a going concern within one year after the date of the consolidated statement of financial position.
There were no adjustments made to reflect the effect of this doubt.
We have
incurred significant operating losses since inception and expect to continue to incur significant operating losses for the foreseeable
future and may never become profitable. The losses result primarily from our conduct of research and development activities. As previously
discussed, we have discontinued further development of our iNKT sponsored trial and paused further patient accrual to our adenosine program
in order to preserve cash resources. Additionally, during the fourth quarter of Fiscal 2024, we sold our shares in Intensity on Nasdaq.
We historically
have funded our operations principally from proceeds from issuances of equity and debt securities. We will require significant additional
capital to make the investments we need to execute our longer-term business plan. Our ability to successfully raise sufficient funds
through the sale of debt or equity securities when needed is subject to many risks and uncertainties and, future equity issuances would
result in dilution to existing shareholders and any future debt securities may contain covenants that limit our operations or ability
to enter into certain transactions.
As
of the date of this filing, we currently anticipate that current cash and cash equivalents is only sufficient to cover our operating
needs through January 2025. These factors raise substantial doubt about our ability to continue as a going concern within one year after
the date of the consolidated statement of financial position.
Cash Flows From Operating
Activities
During
the Fiscal 2025 Six Months, we used cash of $3.25 million to fund operating activities, compared to $7.7 million used during the Fiscal
2024 Six Months. The decrease of $4.5 million period over period is primarily due to our decision to discontinue and pause further patient
accrual in the respective clinical programs in the latter half of Fiscal 2024.
Cash Flows From Investing
Activities
There were
no cash flows from investing activities during both the Fiscal 2025 Six Months and the Fiscal 2024 Six Months.
Cash Flows From Financing
Activities
During the
Fiscal 2025 Six Months, we used net cash of $0.018 million from financing activities. During the Fiscal 2024 Six Months, we generated
cash of $0.6 million from financing activities primarily
attributable to the net proceeds from sales under the ATM program partially offset by the
repayment of lease liability.
Key
Contractual Obligations
Details
of contractual obligations, commitments and contingent liabilities are provided in Note 12, “Commitments and Contingent Liabilities,”
to the unaudited condensed consolidated interim financial statements for the three and six months ended September 30, 2024.
Master Services Agreement
Effective
March 15, 2022, through iOx, we entered into a Master Services Agreement (the “MSA”) with Parexel under which Parexel agreed
to act as clinical service provider (“CRO”) pursuant to a work order (“Work Order”) effective June 1, 2022. Pursuant
to such Work Order, Parexel planned to operate a Phase 2 trial of IMM60 and pembrolizumab in advanced melanoma and NSCLC. The MSA provided
for a five-year term, and the Work Order provided for a term to end upon the completion of the services required. The budget provided
for service fees and pass-through expenses and clinical sites totaling $11.5 million. During Fiscal 2023, we executed two change orders
resulting in a $0.6 million increase in the overall estimated budgeted costs. As a result of our decision to discontinue the development
with respect to this program, on December 20, 2023, we provided Parexel notice of termination of the contract, with a planned termination
date of April 18, 2024. As the ongoing CRO services were wound down, we noticed that expenses incurred under the CRO agreement were higher
than originally budgeted. Parexel agreed to refund $0.552 million to us and release the liability for amounts invoiced and unbilled services
totaling $1.486 million, and we forfeited advanced payments of $1.091 million. As a result, we recognized a net gain on of $0.946 million
as of September 30, 2024. We received the refund from Parexel on October 3, 2024.
Clinical Service Agreement
On March
1, 2023, we, through Tarus, entered into a clinical service agreement with Fortrea Inc. (formerly Labcorp Drug Development Inc.), a third-
party CRO. The term of the agreement is through the earlier of August 14, 2025, or the completion of provision of services and the payment
of contractual obligations. The budgeted costs for the services to be provided is approximately $12.1 million. Because of our decision
to discontinue and pause further accrual of all clinical studies, we are negotiating a revision to the services required under the change
in circumstances.
iOx (iNKT) License
On July
1, 2015, iOx entered into a licensing agreement with Ludwig Institute for Cancer Research Ltd. (“LICR”), which covers certain
technology, intellectual property and know-how and development with respect to iNKT cell agonists to treat human diseases. Under the
terms of the licensing agreement (“LICR License”), LICR granted to iOx an exclusive worldwide license, with the right to
grant sublicenses, under the Licensed Patent and Licensed Technology, each as defined in the LICR License, in each case, to develop,
make, have made, use, sell, offer for sale and import Licensed Products, as defined in the LICR License, subject to certain rights retained
by LICR for academic and research purposes. The LICR License provides for a royalty term of ten years after the first commercial sale,
on a Licensed Product by Licensed Product, country by country basis. Upon the expiration of the applicable royalty term, the license
with respect to such Licensed Product in such country will convert to a non-exclusive, fully paid-up license.
LICR is
entitled to 15,000 GBP as an annual license fee on each annual anniversary of the effective date of the LICR License until royalties
become duly payable and 15,000 GBP as a patent reimbursement fee until LICR has been fully reimbursed for all patent costs incurred prior
to the LICR License.
Additionally,
LICR is entitled to milestone payments totaling up to 20.45 million GBP based upon the first Licensed Product achieving specific clinical,
regulatory and sales-based milestones. LICR is also entitled to milestone payment totaling up to 10.25 million GBP based upon a second
Licensed Product achieving specific clinical, regulatory and sales-based milestones.
Finally,
LICR is entitled to a low-single digit royalty on net sales of Licensed Products that marginally escalates upon sales levels all determined
by territory. LICR is also entitled to a percentage of any sublicensing income that gradually decreases based on the stage of development
of the most advanced Licensed Product that is the subject of the applicable sublicense agreement.
Pursuant
to the terms and conditions of the LICR License, LICR is responsible for managing the preparation, filing, prosecution and maintenance
of all Licensed Patent Rights, as defined in the LICR License. iOx will reimburse LICR for all reasonable patent costs it incurs after
the effective date of the LICR License. Further, the LICR License provides that both parties have the right to termination for material
breach or default in the performance of obligations under the LICR License by the other party and in the event of insolvency of the other
party.
Tarus
(adenosine) License
On July
1, 2022, we acquired Tarus Therapeutics, Inc. Pursuant to the license agreement entered into by Tarus Therapeutics, Inc. and Impetis
Biosciences Limited (“Impetis”) dated October 29, 2019 (“Impetis License”), Impetis granted to Tarus an exclusive
sublicensable worldwide license to develop and commercialize the adenosine receptor antagonists for all indications and certain other
assets which were granted upon exercise of a call option on November 5, 2020.
Under the
terms of the Impetis License, Impetis is eligible to receive payments totaling up to $38 million on an Impetis Compound (as defined in
the Impetis License) based upon achievement of certain clinical and commercial milestones. Milestone payments due in the amount of USD
$1 million for achievement of certain regulatory milestones were paid in July 2022 and a $0.5 million milestone was paid upon dosing
the first patient in September 2023.
Additionally,
commencing upon the First Commercial Sale (as defined in the Impetis License) of a Licensed Product (as defined in the Impetis License),
Impetis is entitled to royalties on worldwide net sales that begin in the mid-single digits and escalate through multiple tiers, with
net sales over $1 billion receiving low double digit royalties.
Pursuant
to the terms and conditions of the Impetis License, Tarus has exclusive and full authority to manage all intellectual property (whether
licensed or not) underlying the assets covered by the Impetis License and any other aspects related to exploitation, development and
commercialization thereof at its own cost, and Impetis must provide Tarus reasonable assistance as requested at Tarus’ cost and
expense. Further, the Impetis License provides that both parties have the right to termination for material breach by the other party
and in the event that the other party undergoes certain events such as a voluntary winding-up, a liquidation or entry into receivership.
Off-balance
Sheet Arrangements
As of September
30, 2024 and March 31, 2024, we did not have any off-balance sheet arrangements, including any relationships with unconsolidated entities
or financial partnership to enhance perceived liquidity.
Transactions
with Related Parties
Significant
related party transactions are detailed in Note 13, “Related Party Transactions,” to the unaudited condensed consolidated
interim financial statements for the three and six months ended September 30, 2024 included elsewhere in this Report.
Financial
and Derivative Instruments
Our financial
instruments recognized in our condensed consolidated interim statements of financial position consist of the following:
Fair value
estimates are made at a specific point in time, based on relevant market information and information about financial instruments. These
estimates are subject to and involve uncertainties and matters of significant judgment; and therefore, these estimates cannot be determined
with precision. Changes in assumptions could significantly affect these estimates.
The following
table summarizes our financial instruments as of September 30, 2024 and March 31, 2024 (in thousands):
| |
|
September
30, 2024
(Unaudited) |
|
March
31, 2024 |
| |
|
|
Amortized
Cost |
|
|
|
FVTOCI |
|
|
|
FVTPL |
|
|
|
Amortized
Cost |
|
|
|
FVTOCI |
|
|
|
FVTPL |
|
| Financial assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Cash and cash equivalents |
|
$ |
1,764 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
5,028 |
|
|
$ |
— |
|
|
$ |
— |
|
| Prepaid expenses and other current assets |
|
$ |
922 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
2,667 |
|
|
$ |
— |
|
|
$ |
— |
|
| |
|
September 30, 2024
(Unaudited) |
|
March 31, 2024 |
| |
|
|
Amortized
Cost |
|
|
|
FVTPL |
|
|
|
Amortized
Cost |
|
|
|
FVTPL |
|
| Financial liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Accounts payable and accrued liabilities |
|
$ |
845 |
|
|
$ |
— |
|
|
$ |
2,836 |
|
|
$ |
— |
|
| Warrant liability |
|
$ |
— |
|
|
$ |
1,138 |
|
|
$ |
— |
|
|
$ |
1,564 |
|
A summary of our risk exposures
as it relates to financial instruments are reflected below.
Fair value of Financial
Instruments
Our financial assets and liabilities
are comprised of cash and cash equivalents, receivables and investments in equities and public entities, accounts payable and accrued
liabilities, lease liability, and warrant liability.
We classify
the fair value of these transactions according to the following fair value hierarchy based on the amount of observable inputs used to
value the instrument:
|
• |
Level 1 – Values are based on unadjusted
quoted prices available in active markets for identical assets or liabilities as of the reporting date. |
|
• |
Level 2 – Values are based on inputs, including
quoted forward prices for commodities, time value and volatility factors, which can be substantially observed or corroborated in
the marketplace. Prices in Level 2 are either directly or indirectly observable as of the reporting date. |
|
• |
Level 3 – Values are based on prices or
valuation techniques that are not based on observable market data. Investments are classified as Level 3 financial instrument. |
Assessment
of the significance of a particular input to the fair value measurement requires judgment and may affect the placement within the fair
value hierarchy.
Management
has assessed that the fair values of cash and cash equivalents, other receivables and accounts payable approximate their carrying amounts
largely due to the short-term maturities of these instruments.
The following
methods and assumptions were used to estimate their fair values:
Warrant
Liability: The fair value is estimated using a Black-Scholes model and in certain cases, a Monte Carlo simulation (Level 3).
Credit Risk, Liquidity Risk and
Foreign Currency Risk
Our financial
instruments are exposed to certain financial risks: Credit Risk, Liquidity Risk and Foreign Currency Risk.
Credit Risk
Credit risk
is the risk of loss associated with a counterparty’s inability to fulfill its payment obligations. The credit risk is attributable
to various financial instruments, as noted below. The credit risk is limited to the carrying value as reflected in our condensed consolidated
interim statements of financial position.
Cash and cash
equivalents: Cash and cash equivalents comprise cash on hand and amounts invested in underlying treasury and money market funds that
are readily convertible to a known amount of cash with three months or less from date of acquisition and are subject to an insignificant
risk of change in value. As of September 30, 2024 and March 31, 2024, cash equivalents was comprised of a money market account with maturities
less than 90 days from the date of purchase. Cash and cash equivalents are held with major international financial institutions and therefore
the risk of loss is minimal.
Liquidity Risk
Liquidity
risk is the risk that we will encounter difficulty in satisfying financial obligations as they become due.
Our approach
to managing liquidity is to ensure, as far as possible, that we will have sufficient liquidity to meet our liabilities when due, under
both normal and stressed conditions without incurring unacceptable losses or risking harm to our reputation. We hold sufficient cash
and cash equivalents to satisfy current obligations under accounts payable and accruals.
We monitor
our liquidity position regularly to assess whether we have the funds necessary to meet our operating needs and needs for investing in
new projects.
As a biotech
company at an early stage of development and without significant internally generated cash flows, there are inherent liquidity risks,
including the possibility that additional financing may not be available to us, or that actual drug development expenditures may exceed
those planned. The current uncertainty in global capital markets could have an impact on our future ability to access capital on terms
that are acceptable to us. There can be no assurance that required financing will be available to us.
Foreign
Currency Risk
While we
operate in various jurisdictions, substantially all of our transactions are denominated in the U.S. Dollar, except the deferred tax liability
in the U.K. settleable in British pound sterling and the Stimunity Convertible Note receivable settleable in euros.
Use
of Estimates and Judgments
The preparation
of the condensed consolidated interim financial statements in conformity with IFRS requires management to make judgments, estimates and
assumptions that affect the application of accounting policies and the reported amounts of assets, liabilities, income and expenses.
Actual results may differ from these estimates.
Estimates
and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which
the estimates are revised and in any future periods affected.
Significant
areas where estimates are made include valuation of financial instruments (including deferred tax assets and liabilities, warrant liabilities,
research and development costs, contingent consideration assumed and measurement
of share-based compensation. Significant areas where critical judgments are applied include
in-process research and development and warrant liabilities.
New
Accounting Standards, Interpretations and Amendments
We are also
unaware of any applicable but not-yet-adopted standards that are expected to materially affect our financial statements for future periods.
Internal
Control Over Financial Reporting
Our management,
including the CEO and CFO, is responsible for establishing and maintaining adequate internal controls over financial reporting (as defined
in Rule 13a-15(f) under the Exchange Act). Our internal control system was designed to provide reasonable assurance to our management
and our Board regarding the reliability of financial reporting and preparation and fair presentation of published financial statements
for external purposes in accordance with IFRS. Internal control over financial reporting includes those policies and procedures that:
|
1. |
pertain to the maintenance of records that in
reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; |
|
2. |
provide reasonable assurance that transactions
are recorded as necessary to permit preparation of financial statements in accordance with IFRS, and that our receipts and expenditures
are being made only in accordance with authorizations of our management and directors; and |
|
3. |
provide reasonable assurance regarding prevention
or timely detection of unauthorized acquisition, use or disposition of our assets that have a material effect on the financial statements. |
All internal
control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can
provide only reasonable assurance with respect to financial statement preparation and presentation. Because of its inherent limitations,
internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to
future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance
with the policies or procedures may deteriorate.
Management
assessed the effectiveness of our internal control over financial reporting as of September 30, 2024. In making this assessment, it used
the criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the
Treadway Commission. Based on the evaluation under these criteria, management identified material weaknesses in our internal controls
over financial reporting and, as a result, management concluded that our internal control over financial reporting was not effective
as of September 30, 2024.
Management
identified the following material weaknesses in our internal control over financial reporting.
|
• |
Management was unable to perform an effective
risk assessment or monitor internal controls over financial reporting; |
|
• |
Management lacks the number of skilled persons
that it requires given the complexity of the reporting requirements that it has to make, which more specifically include the staff
and expertise to (i) properly segregate duties and perform oversight of work performed and to perform compensating controls over
the finance and accounting functions, (ii) establish and perform fair value estimates or subsequently monitor fluctuations in fair
value estimates, and (iii) apply complex accounting principles, including those relating to business combination accounting, income
taxes, warrant liabilities and fair value estimates; and |
|
• |
There are insufficient written policies and procedures
in place to ensure the correct application of accounting and financial reporting with respect to the current requirements of IFRS
and SEC disclosure requirements, some of which specifically relate to investment accounting and fair value measures, assessment of
in-process R&D assets, share-based payments, carrying amounts of goodwill and intangible assets and business combination accounting. |
Public
Securities Filings
Additional
information, including our annual information in our Annual Report, is filed with the Canadian Securities Administrators at www.sedar.com
and with the SEC at www.edgar.com.
22
Portage Biotech (NASDAQ:PRTG)
Gráfica de Acción Histórica
De Nov 2024 a Dic 2024
Portage Biotech (NASDAQ:PRTG)
Gráfica de Acción Histórica
De Dic 2023 a Dic 2024