Olema Oncology Announces Phase 2 Monotherapy Clinical Results to be Presented at the 2023 ESMO Congress
16 Octubre 2023 - 6:02AM

Olema Pharmaceuticals, Inc. (“Olema” or “Olema Oncology,” Nasdaq:
OLMA), a clinical-stage biopharmaceutical company focused on the
discovery, development and commercialization of targeted therapies
for women’s cancers, today announced an oral presentation on the
Phase 2 monotherapy study results of palazestrant (OP-1250), the
Company’s complete estrogen receptor (ER) antagonist (CERAN) and
selective ER degrader (SERD) for the treatment of metastatic breast
cancer, at the upcoming European Society for Medical Oncology
(ESMO) Congress 2023 in Madrid, Spain, on October 22, 2023.
Details of the ESMO Congress 2023 mini oral session
presentation are:
|
Title: |
Updated results from the phase 1/2 study of OP-1250, an oral
complete estrogen receptor (ER) antagonist (CERAN) and selective ER
degrader (SERD) in patients with advanced or metastatic
ER-positive, HER2-negative breast cancer. |
|
Presentation #: |
382MO |
|
Date: |
Sunday, October 22, 2023 |
|
Time: |
08:55 – 09:00 a.m. CEST (02:55 – 03:00 a.m. ET) |
|
Location: |
Bilbao Auditorium – NCC, IFEMA MADRID |
A copy of the presentation will be made available on Olema’s
website under the Science section when it is presented at the
congress. Abstracts for the posters can be found on the ESMO
Congress 2023 website here.
Company Investor Webcast and Conference
Call
Olema will host a webcast and conference call for analysts and
investors to review data presented at the ESMO Congress 2023 as
well as other ongoing studies on Monday, October 23, 2023, at 8:00
a.m. ET (2:00 p.m. CEST). Lead study author, Dr. Nancy U. Lin,
Associate Chief of the Division of Breast Oncology, Susan F. Smith
Center for Women's Cancers, at the Dana-Farber Cancer Institute in
Boston, MA, will join Olema management for the call. Please
register for the webcast by visiting the Investors & Media
section of Olema’s website at olema.com.
About Olema OncologyOlema Oncology is a
clinical-stage biopharmaceutical company focused on the discovery,
development and commercialization of targeted therapies for women’s
cancers. Olema’s lead product candidate, palazestrant (OP-1250), is
a proprietary, orally-available small molecule with dual activity
as both a complete estrogen receptor (ER) antagonist (CERAN) and a
selective ER degrader (SERD). It is currently being evaluated both
as a single agent in an ongoing Phase 2 clinical trial, and in
combination with CDK4/6 inhibitors (palbociclib and ribociclib) and
a PI3Ka inhibitor (alpelisib), in patients with recurrent, locally
advanced or metastatic ER-positive (ER+), human epidermal growth
factor receptor 2-negative (HER2-) breast cancer. Palazestrant has
been granted FDA Fast Track designation for the treatment of
ER+/HER2- metastatic breast cancer that has progressed following
one or more lines of endocrine therapy with at least one line given
in combination with a CDK4/6 inhibitor. Olema is headquartered in
San Francisco and has operations in Cambridge, Massachusetts. For
more information, please visit us at www.olema.com, or follow us on
Twitter and LinkedIn.
Contact:Geoffrey Mogilner, Vice President, Investor Relations
and Communicationsir@olema.com
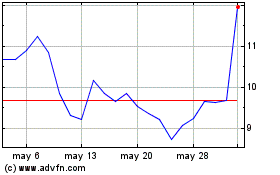
Olema Pharmaceuticals (NASDAQ:OLMA)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
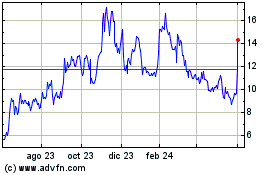
Olema Pharmaceuticals (NASDAQ:OLMA)
Gráfica de Acción Histórica
De May 2023 a May 2024