Edwards’ EVOQUE Valve Replacement System First Transcatheter Therapy to Earn FDA Approval for Tricuspid Valve
02 Febrero 2024 - 6:00AM
Business Wire
Edwards Lifesciences Corporation (NYSE: EW) today announced the
company’s EVOQUE tricuspid valve replacement system is the first
transcatheter therapy to receive U.S. Food and Drug Administration
(FDA) approval for the treatment of tricuspid regurgitation (TR).
The EVOQUE system is indicated for the improvement of health status
in patients with symptomatic severe TR despite optimal medical
therapy (OMT), for whom tricuspid valve replacement is deemed
appropriate by a heart team.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240202805454/en/
(Photo: Edwards Lifesciences)
“Edwards has a long history of leading innovation and pioneering
new therapies to address the unmet needs of patients with
structural heart disease,” said Daveen Chopra, Edwards’ corporate
vice president, transcatheter mitral and tricuspid therapies. “We
are grateful for the strong collaboration with clinicians all over
the world who contributed to the EVOQUE system now being available
through FDA’s Breakthrough Pathway to provide a treatment option to
the many patients in the US suffering with tricuspid valve
disease.”
The EVOQUE system is comprised of a nitinol self-expanding
frame, intra-annular sealing skirt and tissue leaflets made from
the company’s proven bovine pericardial tissue. The EVOQUE valve
will be available in three sizes, all delivered through the same
low-profile transfemoral 28F system.
“Patients suffering with tricuspid regurgitation endure
life-impairing symptoms and, until today, had no approved
transcatheter treatment options,” said Susheel Kodali, MD,
director, Structural Heart and Valve Center at Columbia University
Irving Medical Center/New York-Presbyterian Hospital and TRISCEND
II Study Principal Investigator. “The EVOQUE system is able to
replace the native tricuspid valve, virtually eliminating tricuspid
regurgitation in a wide range of patients. We see significant
improvements in patients’ symptoms and quality-of-life, including
not feeling short of breath and being able to care for themselves,
which ranked highest on a patient preference survey conducted at
baseline with TRISCEND II pivotal trial patients.”
Successful six-month results from the randomized controlled
pivotal trial, TRISCEND II, were presented at TCT 2023 and reported
favorable safety and effectiveness outcomes, demonstrating
superiority to OMT alone and meeting all primary endpoints. Key
findings in the trial included significant reduction or elimination
of tricuspid regurgitation and significant and sustained quality of
life improvement, while demonstrating a favorable balance between
risk and benefit.
In addition to the six-month cohort, 318 of the total 392
randomized patients completed a 1-year visit. The results showed
favorable trends in the device group compared to the control group
in the primary composite endpoints, including all-cause mortality,
tricuspid intervention, heart failure hospitalization, KCCQ, NYHA
and 6MWD. Edwards expects to present the full cohort of 392
TRISCEND II pivotal trial patients at TCT 2024.
The EVOQUE system received CE Mark approval in October 2023,
making it the world’s first transcatheter valve replacement therapy
to receive regulatory approval to treat TR.
About Edwards
Lifesciences
Edwards Lifesciences is the global leader of patient-focused
innovations for structural heart disease and critical care
monitoring. We are driven by a passion for patients, dedicated to
improving and enhancing lives through partnerships with clinicians
and stakeholders across the global healthcare landscape. For more
information, visit Edwards.com and follow us on Facebook,
Instagram, LinkedIn, X and YouTube.
This news release includes forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended,
and Section 21E of the Securities Exchange Act of 1934, as amended.
These forward-looking statements include, but are not limited to,
statements made by Mr. Chopra and Dr. Kodali, and statements
regarding expected product benefits, patient outcomes, objectives
and expectations and other statements that are not historical
facts. Forward-looking statements are based on estimates and
assumptions made by management of the company and are believed to
be reasonable, though they are inherently uncertain and difficult
to predict. Our forward-looking statements speak only as of the
date on which they are made, and we do not undertake any obligation
to update any forward-looking statement to reflect events or
circumstances after the date of the statement. Investors are
cautioned not to unduly rely on such forward-looking
statements.
Forward-looking statements involve risks and uncertainties that
could cause results to differ materially from those expressed or
implied by the forward-looking statements based on a number of
factors as detailed in the company’s filings with the Securities
and Exchange Commission, including its Annual Report on Form 10-K
for the year ended December 31, 2022, and its Quarterly Reports on
Form 10-Q for the quarters ended March 31, June 30, and September
30, 2023. These filings, along with important safety information
about our products, may be found at Edwards.com.
Edwards, Edwards Lifesciences, the stylized E logo, Edwards
EVOQUE, EVOQUE, and TRISCEND are trademarks of Edwards Lifesciences
Corporation. All other trademarks are the property of their
respective owners.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240202805454/en/
Media Contact: Loree Bowen, 714-403-2475 Investor Contact: Mark
Wilterding, 949-250-6826
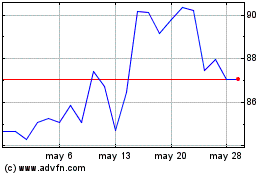
Edwards Lifesciences (NYSE:EW)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
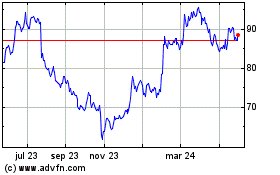
Edwards Lifesciences (NYSE:EW)
Gráfica de Acción Histórica
De May 2023 a May 2024