Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16]
09 Enero 2024 - 8:00AM
Edgar (US Regulatory)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the Month of January 2024 (Report No. 2)
Commission File Number: 001-40303
Inspira Technologies Oxy B.H.N. Ltd.
(Translation of registrant’s name into
English)
2 Ha-Tidhar St.
Ra’anana 4366504, Israel
(Address of principal executive office)
Indicate by check mark whether the registrant
files or will file annual reports under cover of Form 20-F or Form 40-F:
☒ Form 20-F ☐ Form
40-F
CONTENTS
On January 9, 2024,
Inspira Technologies Oxy B.H.N. Ltd., or the Registrant, issued a press release titled “Inspira™ Announces Plans to
Launch and Target Single-Use Disposable Blood Oxygenation Kit for a New Disposable Perfusion Market Opportunity,” a copy of which is furnished as Exhibit 99.1 with this report
of foreign private issuer on Form 6-K.
The
first and second paragraphs and the section titled “Forward-Looking Statements” in the press release are incorporated by
reference into the Registrant’s Registration Statements on Form
F-3 (Registration No. 333-266748) and Form S-8
(Registration No. 333-259057), filed with the Securities and Exchange Commission, to be a part thereof
from the date on which this report is submitted, to the extent not superseded by documents or reports subsequently filed or furnished.
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
Inspira Technologies Oxy B.H.N. Ltd. |
| |
|
|
| Date: January 9, 2024 |
By: |
/s/ Dagi Ben-Noon |
| |
|
Name: |
Dagi Ben-Noon |
| |
|
Title: |
Chief Executive Officer |
2
Exhibit
99.1
Inspira™
Announces Plans to Launch and Target Single-Use Disposable Blood Oxygenation Kit for a New Disposable Perfusion Market Opportunity
The kit is being
designed for the INSPIRA™ ART device series and is intended to be compatible with various life support machines sold in the market
RA’ANANA,
Israel, January 9, 2024 – Inspira
Technologies OXY B.H.N Ltd. (Nasdaq: IINN) (Nasdaq: IINNW) (the “Company” or “Inspira”), a pioneer in life
support technology with a vision to supersede traditional mechanical ventilators, today announced its plan to launch a single-use
disposable blood oxygenation kit (the “Kit”), currently in development, for its INSPIRA ART medical device series. The Kit
is also intended to be compatible with various other life support machines, tapping into the disposable perfusion
systems market.
The Kit,
anticipated to be submitted for US Food & Drug Administration (FDA) authorization in 2025, is a single-use product designed for efficient
blood oxygenation and carbon dioxide removal. The Kit represents a strategic move to enhance hospital budget spending by potentially reducing
costs. The Company is focusing on making the Kit compatible with other medical devices such as certain types of cardio-pulmonary bypass
machines.
Dagi
Ben-Noon, CEO of Inspira Technologies, commented: “Introducing the Kit marks the potential expansion of our market presence and
fast-tracks our goal to deploy our blood oxygenation devices, with potential revenue streams based on the disposable razorblade business
model. We believe that this development, along with the anticipated FDA clearance of our INSPIRA™ ART100
device in the first half of 2024, solidifies our commitment to providing comprehensive and affordable solutions in life support technology.”
Inspira Technologies OXY B.H.N Ltd.
Inspira
Technologies is leading the way in transforming life-support care. Its innovative solutions are paving the way for direct blood oxygenation,
bypassing the lungs, and potentially reducing the need for traditional mechanical ventilation. Beyond this, the Company is committed to
advancing blood circulation technology and incorporating AI-driven monitoring systems. These advancements are part of its strategy to
offer more patient-focused, data-informed care. The integration of these technologies signifies the potential to enhance patient outcomes
and streamline hospital operations, marking a new era in respiratory care.
For more information, please
visit our corporate website:
https://inspira-technologies.com/
Forward-Looking Statement Disclaimer
This press release contains express or implied
forward-looking statements under U.S. Federal securities laws. These forward-looking statements and their implications are based only
on the current expectations of the management of the Company. They are subject to several factors and uncertainties that could cause results
to differ materially from those described in the forward-looking statements. For example, the Company uses forward-looking statements
when it discusses the prospective compatibility of the Kit with other life support machines, the anticipated FDA submission of the Kit
in 2025, that the Kit represents a strategic move to potentially reduce costs, that the Kit fast-tracks the Company’s goal to deploy
its blood oxygenation devices, and the belief that the Kits, along with the anticipated FDA clearance
of its INSPIRA ART device in the first half of 2024, solidifies its commitment to providing comprehensive and affordable solutions
in life support technology. Except as otherwise required by law, the Company undertakes no obligation to publicly release any revisions
to these forward-looking statements to reflect events or circumstances after the date hereof or the occurrence of unanticipated events.
More detailed information about the risks and uncertainties affecting the Company is contained under the heading “Risk Factors”
in the Company’s annual report on Form 20-F for the fiscal year ended December 31, 2022, filed with the SEC, which is available
on the SEC’s website, www.sec.gov.
For more details:
Public Relations Manager
Adi Shmueli
Inspira Technologies
info@inspirao2.com
+972-9-9664485
Copyright © 2018-2024 Inspira Technologies
OXY B.H.N. LTD., All rights reserved.
MRK-ARS-082
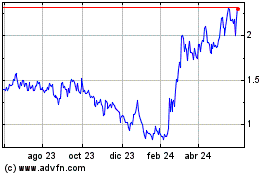
Inspira Technologies Oxy... (NASDAQ:IINN)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
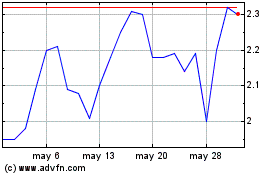
Inspira Technologies Oxy... (NASDAQ:IINN)
Gráfica de Acción Histórica
De May 2023 a May 2024