Avicanna Announces Medical Cannabis Real World Evidence Study through MyMedi.ca
23 Septiembre 2024 - 6:25AM

Avicanna Inc. (“
Avicanna” or the
“
Company) (TSX: AVCN) (OTCQX: AVCNF) (FSE: 0NN) a
biopharmaceutical company focused on the development,
manufacturing, and commercialization of plant-derived
cannabinoid-based products is pleased to announce a large-scale
medical cannabis real world evidence study through the MyMedi.ca
medical cannabis care platform (“Study”). The Study is to be led by
Hance Clarke, MD, FRCPC, PhD. Dr. Clarke is the current President
of The Canadian Pain Society and the Canadian Consortium for the
Investigation of Cannabinoids.
The prospective, non-interventional,
observational study aims to enroll 1,000 patients across the
country to understand the potential therapeutic use of medical
cannabis and potential impact of medical cannabis on pain, sleep,
anxiety, depression, and epilepsy. Utilizing validated
questionnaires, the study seeks to understand the potential impact
of various medical cannabis products and evaluate the change in use
of concomitant medication over a 24-week duration.
“I am pleased that Avicanna has stepped up to
support this important work. We must continue to gather data and
better understand what effects these cannabis products are having
on Canadian patients that are approaching cannabis for therapeutic
benefit” stated Dr. Clarke.
“MyMedi.ca medical cannabis care platform was
established with a number of goals that included, access to medical
cannabis, providing patient support and education, and facilitating
research on cannabinoid medicines,” stated Dr. Karolina Urban,
Executive Vice President Medical Affairs. Dr. Urban further stated,
“We are excited to support the Study and hope that the results of
this study will provide data, evidence, and increase our
understanding of the potential impacts of medical cannabis across a
range of conditions that may facilitate the incorporation of
medical cannabis into standard of care.”
The study was originally initiated by Medical Cannabis by
Shoppers and was part of Avicanna’s commitment to provide
continuation of care to the platforms’ patients but also the
advancement of medical research. Avicanna’s medical cannabis care
platform MyMedi.ca will providing the necessary infrastructure,
patient support, and education for all participating HCPs
nationwide.
Participating patients will have access to the platform’s
products including Avicanna’s own RHO Phyto formulary in addition
to products from select licensed producers that are supporting the
study.
For more information you can go to the website, www.mcrwe.com,
or contact the study coordinator at 416-340-4800 x 4251 or
mcrwe@uhn.ca.
About Avicanna Inc.
Avicanna is a commercial-stage international
biopharmaceutical company focused on the advancement and
commercialization of cannabinoid-based products and formulations
for the global medical and pharmaceutical market segments. Avicanna
has an established scientific platform including R&D and
clinical development leading to the commercialization of more than
thirty proprietary, evidence-based finished products and supporting
four commercial stage business pillars.
- Medical Cannabis formulary (RHO
Phyto™): The formulary offers a diverse range of
proprietary products including oral, sublingual, topical, and
transdermal deliveries with varying ratios of cannabinoids,
supported by ongoing patient, and medical community education. RHO
Phyto is an established leading medical brand in Canada currently
available nationwide to patients across several medical channels
and continues to expand into new international markets.
- Medical cannabis care platform
(MyMedi.ca): MyMedi.ca is a medical cannabis care
platform formed with the aim to better serve medical cannabis
patients’ needs and enhance the patient journey. MyMedi.ca is
operated by Northern Green Canada Inc. and features a diverse
portfolio of products and bilingual pharmacist-led patient support
programs. MyMedi.ca also provides specialty services to distinct
patient groups such as veterans and collaborates with public and
private payers for adjudication and reimbursement. MyMedi.ca
provides educational resources to the medical community to
facilitate the incorporation of medical cannabis into health care
regimens.
- Pharmaceutical products (Trunerox™) and
pipeline: Leveraging Avicanna’s scientific platform,
vertical integration, and real-world evidence, Avicanna has
developed a pipeline of proprietary, indication-specific
pharmaceutical products that are in various stages of clinical
development and commercialization. These cannabinoid-based drug
candidates aim to address unmet medical needs in the areas of
dermatology, chronic pain, and various neurological disorders.
Avicanna’s first indication-specific pharmaceutical drug,
Trunerox™, was approved Q1 2024 by the Health Authority of Colombia
INVIMA as an adjuvant treatment for seizures associated with
Lennox-Gastaut Syndrome and Dravet Syndrome. Trunerox™
has not been approved as a drug in Canada by Health Canada.
- Active pharmaceutical ingredients (Aureus Santa
Marta™): Active pharmaceutical ingredients (“API”)
supplied by the Company’s majority owned subsidiary Santa Marta
Golden Hemp SAS (“SMGH”) is a commercial-stage business dedicated
to providing a various forms high-quality CBD, THC and CBG to the
Company’s international partners for use in the development and
production of food, cosmetics, medical, and pharmaceutical
products. The business unit also forms part of the Company’s supply
chain and is a source of reliable input products for its consumer
retail, medical cannabis, and pharmaceutical products for
globally.
SOURCE Avicanna Inc.
Stay Connected
For more information about Avicanna, visit
www.avicanna.com, contact Ivana Maric by email
at info@avicanna.com or follow us on social media
on LinkedIn, Twitter, Facebook or Instagram.
If you are a Healthcare Professional and would
like to learn more about using medical cannabis in your practice,
please visit Avicenna Academy.
The Company posts updates through videos from
the official Company YouTube channel.
Cautionary Note Regarding
Forward-Looking Information and Statements
This news release contains “forward-looking
information” within the meaning of applicable securities laws.
Forward-looking information contained in this news release may be
identified using words such as, “may”, “would”, “could”, “will”,
“likely”, “expect”, “anticipate”, “believe”, “intend”, “plan”,
“forecast”, “project”, “estimate”, “outlook” and other similar
expressions. Although the Company believes that the expectations
and assumptions on which such forward looking information is based
are reasonable, undue reliance should not be placed on the
forward-looking information because the Company can give no
assurance that they will prove to be correct. Actual results and
developments may differ materially from those contemplated by these
statements. Forward-looking information is subject to a variety of
risks and uncertainties that could cause actual events or results
to differ materially from those projected in the forward-looking
information. Such risks and uncertainties include but are not
limited to current and future market conditions, including the
market price of the common shares of the Company, and the risk
factors set out in the Company’s annual information form dated
April 1, 2024 filed with the Canadian securities regulators and
available under the Company’s profile on SEDAR at www.sedar.com.
The statements in this news release are made as of the date of this
release. The Company disclaims any intent or obligation to update
any forward-looking information, whether as a result of new
information, future events or results or otherwise, other than as
required by applicable securities laws.
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/736fdc04-f148-4bf2-9f80-4dc401159396
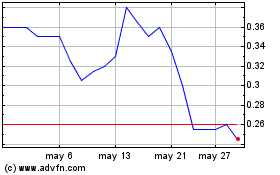
Avicanna (TSX:AVCN)
Gráfica de Acción Histórica
De Nov 2024 a Dic 2024
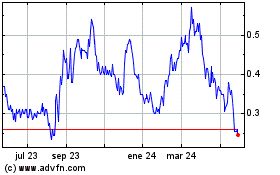
Avicanna (TSX:AVCN)
Gráfica de Acción Histórica
De Dic 2023 a Dic 2024