April 26, 2024
Biodexa Pharmaceuticals
PLC(“Biodexa” or the “Company”)
Biodexa Enters Into Exclusive License to
eRapa™, a Phase 3 Ready Asset for theTreatment of
Familial Adenomatous Polyposis (FAP)
Worldwide rights come with $17 million in
non-dilutive grant funding for Pivotal Phase 3 trial in
FAP
An estimated 100,000 in U.S. and Europe
are afflicted with FAP, precancerous polyps that typically lead to
surgical removal of the colon and/or rectum
In FAP, eRapa holds the potential of
delaying or preventing surgical intervention
Multiple opportunities seen in other
indications, including bladder and prostate cancers
- FAP is a substantially genetic
orphan disease for which there are no approved therapeutic options;
the current standard of care is surveillance and surgery
- Phase 3 FAP program is supported by
a $17 million grant awarded from the Cancer Prevention and Research
Institute of Texas (“CPRIT”) in a competitive process
- Phase 2 results in FAP to be
presented at two leading scientific conferences in Q2 ‘24
- Ongoing Phase 2 study in Non-muscle
Invasive Bladder Cancer expected to read-out in Q2 ‘25
- Phase 2 study in NMI Bladder Cancer
supported by $3 million grant from National Cancer Institute, part
of the National Institutes of Health
Biodexa Pharmaceuticals PLC, (Nasdaq: BDRX), an
acquisition-focused clinical stage biopharmaceutical company
developing a pipeline of innovative products for the treatment of
diseases with unmet medical needs, announced it entered into a
definitive agreement with Rapamycin Holdings Inc. (d/b/a Emtora
Biosciences) (“Emtora”) for the rights to eRapa under an exclusive,
worldwide license (with the ability to grant sublicenses) to
develop, manufacture, commercialize and otherwise advance the
clinical potential of eRapa.
Stephen Stamp, CEO and CFO of Biodexa said,
“Acquiring a Phase 3 ready asset, particularly one supported by $17
million of non-dilutive grant funding, significantly advances
Biodexa’s oncology pipeline and adds numerous valuation catalysts
for our stakeholders. We are delighted to be working with the
Emtora team which has excelled in bringing eRapa close to the end
of Phase 2 in Non-muscle Invasive Bladder Cancer and to the
beginning of a Phase 3 trial in FAP, a devastating disease for
which there is currently no approved pharmacological agent for
altering its progression. Left untreated, it almost always leads to
incredibly invasive surgery and a major deterioration in the
quality of life.”
Stephen Dufilho, Executive Chairman of Emtora
added, “The transaction with Biodexa is the culmination of a
decade-long effort to advance our potentially game-changing eRapa
to a registrational Phase 3 trial - and ultimately to patients in
need. Our story began in San Antonio – where eRapa was originally
invented at the University of Texas and funded in part by grants
from the Cancer Prevention Research Institute of Texas. We thank
the scientists, clinicians, investors and biotech executives that
supported our efforts to reach this important milestone. We look
forward to working with the Biodexa team as we embark upon this
next chapter.”
About eRapaeRapa is a
proprietary oral tablet formulation of rapamycin, also known as
sirolimus. Rapamycin is an mTOR (mammalian Target Of Rapamycin)
inhibitor. mTOR has been shown to have a significant role in the
signalling pathway that regulates cellular metabolism, growth and
proliferation and is activated during tumorgenesis1. Rapamycin is
approved in the US for organ rejection in renal transplantation as
Rapamune®(Pfizer). Through the use of nanotechnology and pH
sensitive polymers, eRapa is designed to address the poor
bioavailability, variable pharmacokinetics and toxicity generally
associated with the currently available forms of rapamycin. eRapa
is protected by a number of issued patents which extend through
2035, with other pending applications potentially providing further
protection beyond 2035.
eRapa in FAPFAP is
characterized as a proliferation of polyps in the colon and/or
rectum, usually occurring in mid-teens. There is no approved
therapeutic option for treating FAP patients, for whom active
surveillance and surgical resection of the colon and/or rectum
remain the standard of care. If untreated, FAP typically leads to
cancer of the colon and/or rectum. There is a significant
hereditary component to FAP with a reported incidence of one in
5,000 to 10,000 in the US2 and one in 11,300 to 37,600 in Europe3.
eRapa has received Orphan Designation in the US with plans to seek
such designation in Europe. Importantly, mTOR has been shown to be
over-expressed in FAP polyps – thereby underscoring the rationale
for using a potent and safe mTOR inhibitor like eRapa to treat
FAP.
Emtora is currently completing an open-label,
multi-center Phase 2 study in 30 patients with confirmed FAP with
the primary endpoints of safety and tolerability, and percentage
change in polyp burden after six months of treatment with eRapa.
The Phase 2 study was partially funded by a $3.0 million grant from
CPRIT.
The results of the Phase 2 study will be
presented at two leading scientific conferences in Q2 24. Following
a positive end of Phase 2 meeting with the FDA and, Emtora plans to
initiate a Phase 3 multi-center, double-blind, placebo-controlled
study in FAP. The Phase 3 study, which is expected to be
registrational, plans to recruit approximately 140 patients across
thirty or more sites, with a primary endpoint being time to a
progression free survival event (the occurrence of which is related
to the reduction in polyp burden studied in the earlier Phase 2
trial). The study is expected to recruit over 15 months and is
supported by a further non-dilutive grant of $17.0 million from
CPRIT.
eRapa in Non-Muscle Invasive Bladder
CancerNon-muscle Invasive Bladder Cancer (“NMIBC”) refers
to tumors found in the tissue that lines the inner surface of the
bladder. The most common treatment is transurethral resection of
the bladder tumor followed by intravesical Bacillus Calmette-Guerin
(“BCG”) with chemotherapy depending upon assessment of risk of
recurrence. NMIBC is the fourth most common cancer in men with an
incidence of 10.1 per 100,000 and 2.5 per 100,000 in women4.
Emtora’s ongoing multi-center, double-blind,
placebo-controlled Phase 2 study in NMIBC is expected to enroll up
to 166 patients with primary endpoints of safety/tolerability and
relapse free survival after 12 months of treatment. The Phase 2
study, which is supported by a $3.0 million non-dilutive grant from
the National Cancer Institute, part of the National Institutes of
Health, is expected to read out in Q2 ’25.
Other Potential Indications for
eRapaA number of rare/orphan gastro-intestinal diseases
(other than FAP) have been identified that share the strong
scientific rationale (relating to mTOR inhibition) and support the
potential utility of eRapa in FAP. Biodexa intends to evaluate such
opportunities over the coming months, including by leveraging
low-cost, investigator-sponsored trials to produce initial proof of
concept data.
The License TransactionThe
transaction terms include the issuance to Emtora of 378,163 of the
Company’s American Depository Shares (representing 5% of the
Company’s issued and outstanding ordinary shares on a fully-diluted
basis (including in-the-money warrants)) at close. In addition, the
Company may pay up to $41.5 million in sales milestones within the
first six months of the first commercial sale of a first-approved
indication of eRapa in major markets, with decreasing milestones
for subsequent approvals for additional indications. The Company is
also obligated to pay single digit tiered royalties on net sales of
eRapa™ in addition to meeting Emtora’s legacy royalty obligations
and paying Emtora a certain (stage-dependent) percentage of income
derived from sublicensing and partnering of eRapa.
Ladenburg Thalmann & Co. Inc. acted as
financial advisor to BioDexa in connection with this
transaction.
The Cancer Prevention and Research
Institute of TexasTo date, CPRIT has awarded $2.9
billion in grants to Texas research institutions and
organizations through its academic research, prevention and product
development research programs. CPRIT has recruited 237
distinguished researchers, supported the establishment, expansion
or relocation of 43 companies to Texas and generated
over $5.7 billion in additional public and private
investment. CPRIT funding has advanced scientific and clinical
knowledge and provided 7.4 million life-saving cancer prevention
and early detection services reaching Texans from all 254 counties.
On November 5, 2019, Texas voters overwhelmingly
approved a constitutional amendment to provide an
additional $3 billion to CPRIT for a total $6
billion investment in cancer research and prevention. Learn
more at https://cprit.texas.gov/.
1. Tian et al., mTOR
Signaling in Cancer and mTOR Inhibitors in Solid Tumor Targeting
Therapy, Int J Mol Sci. 2019 Feb; 20(3):
7552. www.rarediseases.org3. www.orpha.net4. Cassell
et al., World J Oncol. 2019 Jun; 10(3): 123–131
For more information, please contact:
|
Biodexa Pharmaceuticals PLC |
|
Stephen Stamp, CEO, CFOTel: +44 (0)29 20480
180www.biodexapharma.com |
|
|
|
Emtora Biosciences Mark Horsey, CFOTel: +1 210 381
2486www.emtorabio.com |
About Biodexa Pharmaceuticals PLC
Biodexa Pharmaceuticals PLC (listed on NASDAQ:
BDRX) is a clinical stage biopharmaceutical company developing a
pipeline of innovative products for the treatment of diseases with
unmet medical needs. The Company’s lead development programmes
include tolimidone, under development as a novel agent for the
treatment of type 1 diabetes and MTX110, which is being studied in
aggressive rare/orphan brain cancer indications, and
Tolimidone is an orally delivered, potent and
selective inhibitor of lyn kinase. Lyn is a member of the Src
family of protein tyrosine kinases, which is mainly expressed in
hematopoietic cells, in neural tissues, liver, and adipose tissue.
Tolimidone demonstrates glycemic control via insulin sensitization
in animal models of diabetes and has the potential to become a
first in class blood glucose modulating agent.
MTX110 is a solubilised formulation of the
histone deacetylase (HDAC) inhibitor, panobinostat. This
proprietary formulation enables delivery of the product via
convection-enhanced delivery (CED) at chemotherapeutic doses
directly to the site of the tumour, by-passing the blood-brain
barrier and potentially avoiding systemic toxicity.
Biodexa is supported by three proprietary drug
delivery technologies focused on improving the bio-delivery and
bio-distribution of medicines. Biodexa’s headquarters and R&D
facility is in Cardiff, UK. For more information visit
www.biodexapharma.com.
About Emtora
Emtora Biosciences is a clinical stage
biopharmaceutical company headquartered in San Antonio, Texas. The
company is developing eRapaTM, amicro-encapsulated formulation of
the previously approved rapamycin, for the treatment of rare
genetic diseases and cancer. Emtora’s lead indication is Familial
Adenomatous Polyposis. The ubiquitous mTOR protein, involved in
multiple signaling pathways, is overexpressed in FAP polyps.
eRapaTM is a potent mTOR inhibitor believed to provide several
significant advantages over rapamycin including: targeted delivery
to the site of active disease (in addition to systemic exposure);
reduced toxicity and improved tolerability; consistent
pharmacokinetics (potentially eliminating the need for drug level
monitoring) and improved bioavailability. eRapaTM was
originally developed at University of Texas Health San Antonio and
is currently the subject of two ongoing and grant-funded Phase 2
trials. For more information, please
visit www.emtorabio.com.
Forward-Looking Statements
Certain statements in this announcement may
constitute “forward-looking statements” within the meaning of
legislation in the United Kingdom and/or United States. Such
statements are made pursuant to the safe harbor provisions of the
Private Securities Litigation Reform Act of 1995 and are based on
management’s belief or interpretation. All statements contained in
this announcement that do not relate to matters of historical fact
should be considered forward-looking statements. In certain cases,
forward-looking statements can be identified by the use of words
such as “plans”, “expects” or “does not anticipate”, or “believes”,
or variations of such words and phrases or statements that certain
actions, events or results “may”, “could”, “would”, “might” or
“will be taken”, “occur” or “be achieved.” Examples of
forward-looking statements include, among others, statements we
make regarding the assignment of rights to eRapa and related
license from Emtora and the Company’s ability to close the
transaction, potential uses by the Company of eRapa, information
related to clinical trials, and potential benefits of
eRapa.Forward-looking statements and information are subject to
various known and unknown risks and uncertainties, many of which
are beyond the ability of the Company to control or predict, that
may cause their actual results, performance or achievements to be
materially different from those expressed or implied thereby, and
are developed based on assumptions about such risks, uncertainties
and other factors set out herein.
Reference should be made to those documents that
Biodexa shall file from time to time or announcements that may be
made by Biodexa in accordance with the rules and regulations
promulgated by the SEC, which contain and identify other important
factors that could cause actual results to differ materially from
those contained in any projections or forward-looking statements.
These forward-looking statements speak only as of the date of this
announcement. All subsequent written and oral forward-looking
statements by or concerning Biodexa are expressly qualified in
their entirety by the cautionary statements above. Except as may be
required under relevant laws in the United States, Biodexa does not
undertake any obligation to publicly update or revise any
forward-looking statements because of new information, future
events or events otherwise arising.
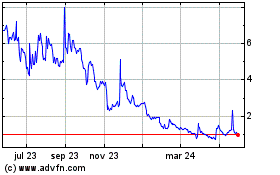
Biodexa Pharmaceuticals (NASDAQ:BDRX)
Gráfica de Acción Histórica
De Abr 2024 a May 2024
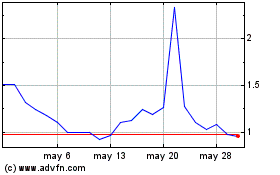
Biodexa Pharmaceuticals (NASDAQ:BDRX)
Gráfica de Acción Histórica
De May 2023 a May 2024