Crossject achieves key ZEPIZURE® manufacturing milestone
18 Julio 2024 - 12:30AM

- Successful completion of an
additional Registration Batch of ZEPIZURE® at new manufacturing
site
- Milestone complements the satisfying
results from previous batches under stability studies, and is part
of the positive manufacturing data generated since 2021
- Manufacturing progress supports
timelines toward granting of Emergency Use Authorization (EUA)
Dijon, France, July 18, 2024, 07:30 CET
-- Crossject (ISIN: FR0011716265; Euronext: ALCJ), a specialty
pharma company in advanced phases of development and registration
for ZEPIZURE®, its emergency treatment
for the management of epileptic crises based on its award-winning
needle-free auto-injector ZENEO®, announces the successful
completion of a new registration batch of ZEPIZURE®, at Eurofins’
qualified facility, which is designated to be the CDMO partner that
will ensure fill and finish activities for the deliveries to the
U.S. Biomedical Advanced Research and Development Authority
(BARDA).
This milestone follows a previous batch produced
on the same filling equipment in December 2023, which delivered
good results. It is part of the ZEPIZURE® manufacturing program,
which includes several batches from its commercial site, aimed at
confirming, among other parameters, the shelf-life of ZEPIZURE®.
These new advances are in line with previous long-term stability
data.
Manufacturing batches are critical parts of the
dossier that will be submitted to the U.S. Food and Drug
Administration (FDA) to support ZEPIZURE®’s application for EUA.
Combined with our upcoming U.S. bioequivalence study, they will
also form the bedrock of our future New Drug Application filings
from 2025 onward.
Completion of this registration batch now paves
the way for gathering new data, which will further demonstrate the
quality of the products manufactured with the new equipment and
ensure their satisfactory regulatory standing for widespread
commercial use, starting with its delivery to the U.S. Government
for national preparedness. Crossject is targeting its next
milestones with the FDA regarding EUA of ZEPIZURE® for status
epilepticus seizures in Q1 2025.
“We are very pleased with the progress in
setting-up a new CDMO partner, increasing fill-and-finish
capabilities for ZENEO®. The successful production of this
registration batch, as well as the earlier batches, are a testament
to the hard work and dedication of both teams over the last 18
months. Beyond ZEPIZURE®, these achievements in our manufacturing
process, regulatory standing and industrial scale-up truly have
strategic value as a prelude to a broad deployment of our ZENEO®
technology in epilepsy and in our other targeted market
opportunities,” said Patrick Alexandre, CEO of
Crossject.
About Crossject
Crossject SA (Euronext:
ALCJ; www.crossject.com) is an emerging specialty
pharmaceuticals company developing medicines for emergency
situations harnessing its award-winning needle-free auto-injector
ZENEO® platform. Crossject is in advanced regulatory development
for ZEPIZURE®, an epileptic rescue therapy, for which it has a $60
million contract* with the U.S. Biomedical Advanced Research and
Development Authority (BARDA). The Company’s versatile ZENEO®
platform is designed to enable patients or untrained caregivers to
easily and instantly deliver a broad range of emergency drugs via
intramuscular injection on bare skin or even through clothing. The
Company’s other products in development include mainly solutions
for allergic shocks and adrenal insufficiencies, as well as
therapies and other emergency indications.
* Contract no: 75A50122C00031 with the
Department of Health and Human Services; Administration for
Strategic Preparedness and Response; Biomedical Research and
Development Authority
For further information, please contact:
|
Investors Natasha DrapeauCohesion Bureau+41 76 823
75 27natasha.drapeau@cohesionbureau.com |
MediaSophie BaumontCohesion Bureau+33 6 27 74 74
49sophie.baumont@cohesionbureau.com |
|
Crossject (EU:ALCJ)
Gráfica de Acción Histórica
De Sep 2024 a Oct 2024
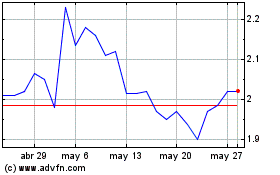
Crossject (EU:ALCJ)
Gráfica de Acción Histórica
De Oct 2023 a Oct 2024