NEW
YORK, July 30, 2024 /PRNewswire/ -- The
global clinical trial management system (CTMS) market size is
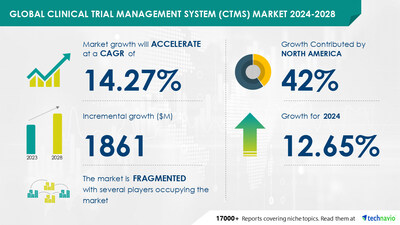
estimated to grow by USD 1.86 billion
from 2024-2028, according to Technavio. The market is estimated to
grow at a CAGR of almost 14.27% during the forecast period.
Increasing healthcare expenditure is driving market
growth, with a trend towards increasing outsourcing of
clinical trial process. However, rising cost of clinical
trials poses a challenge. Key market players include Advarra Inc.,
ArisGlobal LLC, Clinion Inc., Crucial Data Solutions Inc., Dassault
Systemes SE, DATATRAK International Inc., DSG Inc., Ennov SAS,
eResearchTechnology GmbH, International Business Machines Corp.,
Laboratory Corp. Of America Holdings, MasterControl Solutions Inc.,
Medfiles USA, MedNet, Oracle
Corp., Parexel International Corp., PHARMASEAL, RealTime Software
Solutions LLC, Veeva Systems Inc., and Wipro Ltd..
Get a detailed analysis on regions, market
segments, customer landscape, and companies - Click for the
snapshot of this report
|
Forecast
period
|
2024-2028
|
|
Base Year
|
2023
|
|
Historic
Data
|
2018 - 2022
|
|
Segment
Covered
|
Deployment (On-premise
and Cloud), End-user (Pharmaceutical and biotechnology companies,
CROs, and Others), and Geography (North America, Europe, Asia, and
Rest of World (ROW))
|
|
Region
Covered
|
North America, Europe,
Asia, and Rest of World (ROW)
|
|
Key companies
profiled
|
Advarra Inc.,
ArisGlobal LLC, Clinion Inc., Crucial Data Solutions Inc., Dassault
Systemes SE, DATATRAK International Inc., DSG Inc., Ennov SAS,
eResearchTechnology GmbH, International Business Machines Corp.,
Laboratory Corp. Of America Holdings, MasterControl Solutions Inc.,
Medfiles USA, MedNet, Oracle Corp., Parexel International Corp.,
PHARMASEAL, RealTime Software Solutions LLC, Veeva Systems Inc.,
and Wipro Ltd.
|
Key Market Trends Fueling Growth
In the pharmaceutical industry, the shift towards outsourcing
drug discovery processes to subcontract laboratories has been a
significant trend since 2011. This transformation has enabled small
companies to focus on manufacturing and marketing activities while
outsourcing research and testing to external resources. The
increasing popularity of this approach is due to the substantial
cost savings it offers, as companies can outsource these functions
for a fraction of the cost of building and maintaining their own
research facilities. This outsourcing trend is particularly
prevalent in countries like the US, the UK, Germany, and Japan, which have been outsourcing drug
discovery to emerging economies such as India and Brazil. The high volume of data generated
during the drug discovery process necessitates the use of Clinical
Trial Management Systems (CTMS) to manage and analyze this data
effectively. The growing number of tests carried out in subcontract
laboratories has led to a corresponding increase in demand for CTMS
solutions. CTMS plays a crucial role in streamlining the clinical
trial process, ensuring compliance with regulatory requirements,
and facilitating data management and analysis. As the outsourcing
trend continues, the demand for CTMS is expected to grow, providing
opportunities for market expansion.
The Clinical Trial Management System (CTMS) market is
experiencing significant trends that are transforming the clinical
trial landscape. Robust reporting and centralizing recruitment are
key priorities for multinational companies and local CROs.
ERegulatory, eSOURCE, accounting, and aggregate reporting provide
network-wide visibility and oversight. Digitalization, personalized
medicine, outsourcing, and externalization are driving growth in
the market. Robust reporting, decentralized trial models, and evidence-based
facilities are essential for regulatory compliance. ERegulatory,
ERT, and Bioclinica are leading providers in Healthcare IT.
Cloud-based CTMS, telemedicine solutions, and decentralized clinical trials are crucial in
the current COVID-19 era. Costs, system security, backups,
upgrades, and uptime consistency are critical considerations for
CTM systems. AI and machine learning are enhancing trial efficiency
and reducing costs. Vaccines and CTM systems are essential in the
fight against COVID-19. The market is expected to continue growing,
with investment in digitalization and regulatory policies being key
drivers.
Discover 360° analysis of this market. For
complete information, schedule your consultation- Book
Here!
Market Challenges
- Clinical trials are a crucial part of bringing new drugs to
market, but their rising costs are a significant concern for the
pharmaceutical industry. One major challenge is patient enrollment
and retention, which can prolong the trial duration and increase
expenses. According to various studies, the average cost per
patient in the US for clinical trials is approximately USD41,117, with phase 3 trials being the most
expensive, costing around USD20
million. Another factor contributing to the rising costs is
the need for extensive clinical data collection due to new
regulations. This can make trials complex and expensive, especially
for drugs targeting chronic diseases. The longer a trial lasts, the
more it costs, making clinical trial management systems essential
to streamline the process. However, some clinical research
associates may prefer manual work to cut costs, hindering the
growth of the market. To address this issue, increasing patient
awareness is the most feasible solution. By educating patients
about the importance and benefits of clinical trials, the number of
willing participants is likely to rise, reducing the overall cost
and duration of trials. Pharmaceutical companies should focus on
patient education rather than persuasion to ensure a successful
clinical trial process.
- In the dynamic healthcare IT landscape, Clinical Trial
Management Systems (CTMS) have become essential tools for managing
complex clinical trials. However, implementing CTMS comes with
challenges. Decentralized trials and the rise of vaccines
require cloud-based CTMS for flexibility. Telemedicine solutions
and COVID-NMA necessitate robust system security, backups, and
upgrades. Centralization of data, growth, and AI/machine learning
algorithms demand uptime consistency and automation. Clinical trial
managers need access to data on devices like mobile workstations,
laptops, and tablets via online dashboards. Trial planning,
monitoring activities, regulatory procedures, supplies, finance,
and subscription-based software upgrades require scalability.
Add-ons, regulatory norms, document management processes, data
quality, participant safety, and adherence to regulatory procedures
are key considerations. Solutions like Clinion offer IWRS/RTSM,
EDC, eCOA, accruals, deviations, and scalability to address these
challenges.
For more insights on driver and
challenges - Download a Sample Report
Segment Overview
This clinical trial management system (ctms) market report
extensively covers market segmentation by
- Deployment
- End-user
- 2.1 Pharmaceutical and biotechnology companies
- 2.2 CROs
- 2.3 Others
- Geography
- 3.1 North America
- 3.2 Europe
- 3.3 Asia
- 3.4 Rest of World (ROW)
1.1 On-premise- The Clinical Trial Management System
(CTMS) market primarily consists of on-premises and cloud-based
deployment models. On-premises CTMS offers enhanced data security
and control, making it a preferred choice for large firms. However,
the significant investment required for infrastructure and
maintenance poses a challenge for smaller organizations.
Conversely, cloud-based CTMS offers cost-effectiveness and ease of
use. Major on-premises CTMS vendors include Siebel and IMPACT.
Despite the high cost, large firms continue to prefer on-premises
CTMS due to the perceived level of data security. This segment's
growth in the global CTMS market is anticipated to be driven by
these factors during the forecast period.
For more information on market segmentation with geographical
analysis including forecast (2024-2028) and historic data (2018 -
2022) - Download a Sample Report
Research Analysis
The Clinical Trial Management System (CTMS) market is a
significant segment of Healthcare IT, facilitating the planning,
execution, and management of clinical trials. With the increasing
focus on decentralized
clinical trials and the development of vaccines, CTMS solutions
have gained prominence. Cloud-based CTMS is a popular choice due to
its accessibility, scalability, and cost-effectiveness.
Telemedicine solutions and IWRS/RTSM integrate seamlessly with
CTMS, enhancing trial efficiency. EDC, eCOA, accruals, deviations,
and robust reporting are essential features. Outsourcing and
externalization to multinational companies and local CROs are
common practices. Regulatory policies necessitate investment in
CTMS solutions for ensuring compliance. Clario and other advanced
systems offer features like ERT, making trial management more
streamlined.
Market Research Overview
The Clinical Trial Management System (CTMS) market in Healthcare
IT is experiencing significant growth due to the increasing
adoption of decentralized clinical trials, particularly in the
development of vaccines. Cloud-based CTMS solutions are gaining
popularity for their accessibility and cost-effectiveness.
Telemedicine solutions and AI-driven technologies, including
machine learning algorithms and automation, are being integrated
into CTMS to improve efficiency and data quality. Costs, system
security, backups, and upgrades are key considerations for CTMS
implementation. Uptime consistency, centralization of data, and
regulatory norms are crucial for ensuring trial integrity and
participant safety. Clinical trial managers require access to
real-time data from devices such as mobile workstations, laptops,
tablets, and online dashboards for effective trial planning,
monitoring activities, and regulatory procedures. CTMS systems
include modules for supplies, finance, and accounting on a
subscription basis, with software upgrades and add-ons available
for customization. Digitalization of clinical trials through
eRegulatory, eSOURCE, IWRS/RTSM, EDC, eCOA, accruals, deviations,
and scalable, robust reporting is driving growth in the market.
Centralizing recruitment and network-wide visibility are also
important for oversight and personalized medicine initiatives.
Outsourcing and externalization to local CROs and multinational
companies are common practices in the industry. Clario and other
trial models are embracing decentralized approaches and evidence-based
facilities to streamline regulatory policies and investment in the
sector.
Table of Contents:
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation
- Deployment
-
- End-user
-
- Pharmaceutical And Biotechnology Companies
- CROs
- Others
- Geography
-
- North America
- Europe
- Asia
- Rest Of World (ROW)
7 Customer Landscape
8 Geographic Landscape
9 Drivers, Challenges, and Trends
10 Company Landscape
11 Company Analysis
12 Appendix
About Technavio
Technavio is a leading global technology research and advisory
company. Their research and analysis focuses on emerging market
trends and provides actionable insights to help businesses identify
market opportunities and develop effective strategies to optimize
their market positions.
With over 500 specialized analysts, Technavio's report library
consists of more than 17,000 reports and counting, covering 800
technologies, spanning across 50 countries. Their client base
consists of enterprises of all sizes, including more than 100
Fortune 500 companies. This growing client base relies on
Technavio's comprehensive coverage, extensive research, and
actionable market insights to identify opportunities in existing
and potential markets and assess their competitive positions within
changing market scenarios.
Contacts
Technavio Research
Jesse Maida
Media & Marketing Executive
US: +1 844 364 1100
UK: +44 203 893 3200
Email: media@technavio.com
Website: www.technavio.com/
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/clinical-trial-management-system-ctms-market-size-is-set-to-grow-by-usd-1-86-billion-from-2024-2028--increasing-healthcare-expenditure-to-boost-the-market-growth-technavio-302208770.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/clinical-trial-management-system-ctms-market-size-is-set-to-grow-by-usd-1-86-billion-from-2024-2028--increasing-healthcare-expenditure-to-boost-the-market-growth-technavio-302208770.html
SOURCE Technavio