AstraZeneca Safety Analyses for Roxadustat Show Top-Line Results
10 Mayo 2019 - 1:44AM
Noticias Dow Jones
By Oliver Griffin
AstraZeneca PLC (AZN.LN) said Friday that pooled cardiovascular
safety analyses of its global phase 3 program for roxadustat has
produced top-line results.
The pharmaceutical company said the pooled results will inform
discussions with regulatory authorities concerning roxadustat,
which is used to treat anemia in patients with chronic kidney
disease.
AstraZeneca and FibroGen Inc. (FGEN), the treatment's
originator, will now conduct talks with the U.S. Food and Drug
Administration to prepare for regulatory submission.
Roxadustat is expected to be submitted for regulatory approval
in the second half of this year.
Write to Oliver Griffin at oliver.griffin@dowjones.com;
@OliGGriffin
(END) Dow Jones Newswires
May 10, 2019 02:29 ET (06:29 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
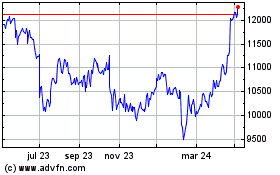
Astrazeneca (LSE:AZN)
Gráfica de Acción Histórica
De Mar 2024 a Abr 2024
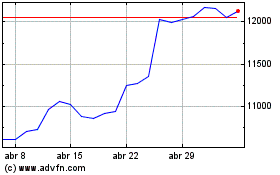
Astrazeneca (LSE:AZN)
Gráfica de Acción Histórica
De Abr 2023 a Abr 2024